
(a)
Interpretation:
The amino acid formed when
Concept introduction:
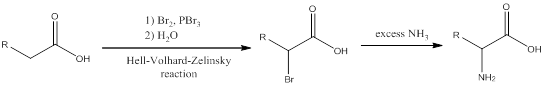
To identify: the amino acid formed when the given carboxylic acid is treated with bromine, phosphorous tribromide and followed by excess ammonia,
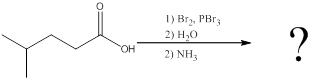
(b)
Interpretation:
The amino acid formed when carboxylic acid is treated with bromine and phosphorous tribromide, followed by water and the resulting
Concept introduction:

To identify: the amino acid formed when the given carboxylic acid is treated with bromine, phosphorous tribromide and followed by excess ammonia,
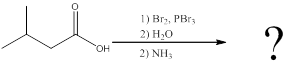
(c)
Interpretation:
The amino acid formed when carboxylic acid is treated with bromine and phosphorous tribromide, followed by water and the resulting
Concept introduction:

To identify: the amino acid formed when the given carboxylic acid is treated with bromine, phosphorous tribromide and followed by excess ammonia,
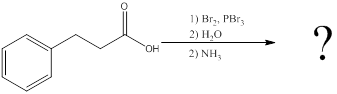
(d)
Interpretation:
The amino acid formed when carboxylic acid is treated with bromine and phosphorous tribromide, followed by water and the resulting
Concept introduction:

To identify: the amino acid formed when the given carboxylic acid is treated with bromine, phosphorous tribromide and followed by excess ammonia,
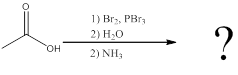
Want to see the full answer?
Check out a sample textbook solution
Chapter 25 Solutions
ORGANIC CHEMISTRY(EBOOK)-W/WILEYPLUS
- A 20.3 mL sample of 0.263 M triethylamine, (C2H5)3N, is titrated with 0.252 M hydrochloric acid. (1) At the titration midpoint, the pH is . (2) At the equivalence point, the pH is .arrow_forwardd. 3,4,5-trimethoxybenzoyl chloride . What is the order of decreasing reactivity towards nucleophilic acyl substitution for the arboxylic acid derivatives? (most reactive first) A. B. 0 0 O 0 0 H3C-C-O-C-CH3 H3C-C-N(CH3)2 H3C-C-OCH 3 (CH3)2CH-C-OCH3 I || ။ IV a. I, II, III, IV b. I, III, IV, II C. II, IV, III, I d. II, I, III, IV 0 0 0 0 0 R-C-O C-R R-C-NH2 R-C OR R-C-CI a. I, III, II, IV | 11 III IV b. II, III, I, IV c. III, II, I, IV d. IV, I, III, IIarrow_forwardB. d. a hydrate 4. Give the major organic product(s) for each of the following reactions or sequences of reactions. Show all relevant stereochemistry [4 ONLY]. A. CH₂OH PCC CH2Cl2 0 H KCN HCN 2arrow_forward
- Propose a synthesis of the anti-inflammatory drug Ibuprofen from benzene. Show all reagents and all intermediate structures. Assume that ortho and para isomers can be separated. (CH3)2CHCH2 CH3 CHCOOH 1buprofen be requiredarrow_forwardAssuming that no equilibria other than dissolution are involved, calculate the molar solubility of each of the following from its solubility product: (a) KHC4H4O6arrow_forwardAnswer the following by equation 1. reactio of CH3MgBr with Acetone [CH3COCH3] 2. acetal formation reaction of acetaldehyde [CH3CHO] 3. preparation of ethylmethylether [C2H5OCH3] 4. the acidity of the carboxylic acid depends and affected by the substitutions on the rest of the acid molecule: draw 2 structures of acids to show the different effects on acidity by different subsarrow_forward
- Consider the reaction sequence below to answer the following questions: 0 0 0 0 0 1. NaOEt, EtOH H3O* OEt OET 2 PhCH Br heat Ph + EtOH + CO₂ CHh B C A A. The starting material A in this reaction sequence is called a a. ẞ-keto ester b. a-carboethoxy ketone C. malonic ester d. acetoacetic ester B. Conversion of A into B is a type of reaction termed a. an acylation b. an enolation C. d. an alkylation a phenylation f reactionsarrow_forward1. Refer to the compounds below to answer the following questions: CO₂Et 0 C. H O O₂N-CH2-C-CH3 0 OEt || 111 A. Indicate all the acidic hydrogens in Compounds I through IV. IV B. Indicate which hydrogens in Compound II are the most acidic. Explain your answer C. Choose the most acidic compound from Compounds I - IV. Explain your choice.arrow_forwardShow how you would accomplish the following transformations. More than one step may be required. ow all reagents and all intermediate structures [one ONLY] A. H Br H CH3 NHz CH3 CH3 B. CH3CH2C-Br CH3CH2C-CN CH3 CH3.arrow_forward
- Show how you would accomplish the following transformations. More than one step may be required. now all reagents and all intermediate structures [one ONLY] A. H Br H CH3 NHz CH3 CH3 B. CH3CH2C-Br CH3 CH3CH2C-CN CH3arrow_forwardCan I please get help with this?arrow_forwardC. I, II, III Consider the reaction sequence below to answer the following questions: 0 0 1. NaOEt, EtOH ΕΙΟ OEt 2 Compound X CO₂Et NaOEt, EtOH CO₂Et Br Compound Y A Compound Z A. Compound X, diethyl propanedioate, is more commonly known as a. ethyl acetoacetate acetoacetic ester b. C. oxalic ester d. malonic ester B. Write the complete stepwise mechanism for the conversion of Compound X into Compound Y. Show all electron flow with arrows and draw all intermediate structures.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





