
ORGANIC CHEMISTRYPKGDRL+MLCRL MDL
3rd Edition
ISBN: 9781119416746
Author: Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 23, Problem 73CP
Interpretation Introduction
Interpretation:
The coupling product obtained by treating the given compound with Grubbs catalyst and the chiral centers present in the compound should be identified.
Concept Introduction:
Grubbs catalyst: This catalyst is used to achieve
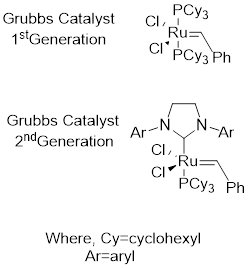
Alkene metathesis: It involves two stages first; the starting material in presence of catalyst forms two possible intermediates. Next the intermediates react with the starting material and results to form product.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Match the denticity to the ligand.
Water
monodentate
✓
C₂O2
bidentate
H₂NCH₂NHCH2NH2 bidentate
x
EDTA
hexadentate
Question 12
Partially correct
Mark 2 out of 2
Flag question
Provide the required information for the coordination compound shown below:
Na NC-Ag-CN]
Number of ligands:
20
Coordination number: 2✔
Geometry: linear
Oxidation state of transition metal ion: +3 x
in 12
correct
out of 2
question
Provide the required information for the coordination compound shown below.
Na NC-Ag-CN]
Number of ligands:
20
Coordination number: 2
Geometry: linear
0
Oxidation state of transition metal ion:
+3X
Can you explain step by step behind what the synthetic strategy would be?
Please explain step by step in detail the reasoning behind this problem/approach/and answer. thank you!
Chapter 23 Solutions
ORGANIC CHEMISTRYPKGDRL+MLCRL MDL
Ch. 23.1 - Identify which of the following reagents is...Ch. 23.2 - Prob. 2CCCh. 23.2 - Prob. 3CCCh. 23.2 - Prob. 4CCCh. 23.2 - Prob. 5CCCh. 23.2 - Prob. 6CCCh. 23.3 - Show how you would prepare 1-butylcyclopentene...Ch. 23.3 - Prob. 7PTSCh. 23.3 -
Using any two organohalides of your choice...Ch. 23.3 - Prob. 9ATS
Ch. 23.4 - Prob. 10CCCh. 23.4 - Prob. 11CCCh. 23.5 - Prob. 2LTSCh. 23.6 - Prob. 3LTSCh. 23.6 - Prob. 16PTSCh. 23.6 - Prob. 17PTSCh. 23.6 - Prob. 18ATSCh. 23.6 - Prob. 19ATSCh. 23.7 - Prob. 4LTSCh. 23.7 - Prob. 20PTSCh. 23.7 - Prob. 21PTSCh. 23.7 - Prob. 22ATSCh. 23.7 - Prob. 23ATSCh. 23.8 - Prob. 5LTSCh. 23.8 - Prob. 24PTSCh. 23.8 - Prob. 25ATSCh. 23.8 - Prob. 26ATSCh. 23.9 - Prob. 27CCCh. 23.9 - Prob. 28CCCh. 23.9 - Prob. 29CCCh. 23.9 - Prob. 6LTSCh. 23.9 - Prob. 30PTSCh. 23.9 - Prob. 31PTSCh. 23.9 - Prob. 32ATSCh. 23 - Prob. 33PPCh. 23 - Prob. 34PPCh. 23 - Prob. 35PPCh. 23 - Prob. 36PPCh. 23 - Prob. 37PPCh. 23 - Prob. 38PPCh. 23 - Prob. 39PPCh. 23 - Prob. 40PPCh. 23 - Prob. 41PPCh. 23 - Prob. 42PPCh. 23 - Prob. 43PPCh. 23 - Prob. 44PPCh. 23 - Prob. 45PPCh. 23 - Prob. 46PPCh. 23 - Using 1-pentene as your only source of carbon...Ch. 23 - Prob. 48PPCh. 23 - Prob. 49PPCh. 23 - Prob. 50PPCh. 23 - Prob. 51PPCh. 23 - Prob. 52PPCh. 23 - Prob. 53PPCh. 23 - Prob. 54PPCh. 23 - Prob. 55PPCh. 23 - Prob. 56PPCh. 23 - Prob. 57PPCh. 23 - Prob. 58PPCh. 23 - Prob. 59IPCh. 23 - Prob. 60IPCh. 23 - Prob. 61IPCh. 23 - Prob. 62IPCh. 23 - Prob. 64IPCh. 23 - Prob. 66IPCh. 23 - Prob. 68IPCh. 23 - Prob. 69IPCh. 23 - Prob. 70IPCh. 23 - Prob. 71CPCh. 23 - Prob. 72CPCh. 23 - Prob. 73CPCh. 23 - Prob. 74CPCh. 23 - Prob. 75CPCh. 23 - Prob. 76CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2. Predict the product(s) that forms and explain why it forms. Assume that any necessary catalytic acid is present. .OH HO H₂N OHarrow_forwardconsider the rate of the reaction below to be r. Whats the rate after each reaction? Br + NaCN CN + NaBr a. Double the concentration of alkyl bromide b. Halve the concentration of the electrophile & triple concentration of cyanide c. Halve the concentration of alkyl chloridearrow_forwardPredict the organic reactant that is involved in the reaction below, and draw the skeletal ("line") structures of the missing organic reactant. Please include all steps & drawings & explanations.arrow_forward
- What are the missing reagents for the spots labeled 1 and 3? Please give a detailed explanation and include the drawings and show how the synthesis proceeds with the reagents.arrow_forwardWhat is the organic molecule X of the following acetal hydrolysis? Please draw a skeletal line structure and include a detailed explanation and drawing of how the mechanism proceeds. Please include any relevant information that is needed to understand the process of acetal hydrolysis.arrow_forwardWhat are is the organic molecule X and product Y of the following acetal hydrolysis? Please draw a skeletal line structure and include a detailed explanation and drawing of how the mechanism proceeds. Please include any relevant information that is needed to understand the process of acetal hydrolysis.arrow_forward
- At 300 K, in the decomposition reaction of a reactant R into products, several measurements of the concentration of R over time have been made (see table). Without using graphs, calculate the order of the reaction. t/s [R]/(mol L-1) 0 0,5 171 0,16 720 0,05 1400 0,027arrow_forwardPredict the organic products that form in the reaction below, and draw the skeletal ("line") structures of the missing organic products. Please include all steps & drawings & explanations.arrow_forwardWhat are the missing reagents for the spots labeled 1 and 3? Please give a detailed explanation and include the drawings and show how the synthesis proceeds with the reagents.arrow_forward
- What are the products of the following acetal hydrolysis? Please draw a skeletal line structure and include a detailed explanation and drawing of how the mechanism proceeds. Please include any relevant information that is needed to understand the process of acetal hydrolysis.arrow_forwardWhat would happen if you added the HCI to the Grignard reagent before adding benzophenone? Draw a reaction mechanism to support your answer.arrow_forwardAt 300 K, in the decomposition reaction of a reactant R into products, several measurements of the concentration of R over time have been made (see table). Calculate the order of the reaction. t/s [R]/ (mol L-1) 0 0,5 171 0,16 720 0,05 1400 0,027arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License