
CHEM: STRUC & PROP (LL) >CUST PKG<
2nd Edition
ISBN: 9780137390977
Author: Tro
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 94E
Determine whether each pair of structures are isomers or the same molecule drawn in two different ways.
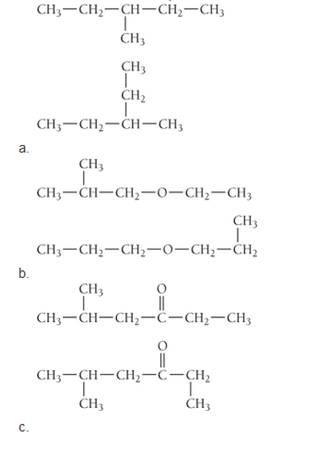
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Calculate the free energy of formation of 1 mol of Cu in cells where the electrolyte is 1 mol dm-3 Cu2+ in sulfate solution, pH 0. E° for the Cu2+/Cu pair in this medium is +142 mV versus ENH.Assume the anodic reaction is oxygen evolution.Data: EH2 = -0.059 pH (V) and EO2 = 1.230 - 0.059 pH (V); 2.3RT/F = 0.059 V
If the normal potential for the Fe(III)/Fe(II) pair in acid at zero pH is 524 mV Hg/Hg2Cl2 . The potential of the saturated calomel reference electrode is +246 mV versus the NHE. Calculate E0 vs NHE.
Given the galvanic cell whose scheme is: (-) Zn/Zn2+ ⋮⋮ Ag+/Ag (+). If we know the normal potentials E°(Zn2+/Zn) = -0.76V and E°(Ag+/Ag) = 0.799 V. Indicate the electrodes that are the anode and the cathode and calculate the E0battery.
Chapter 21 Solutions
CHEM: STRUC & PROP (LL) >CUST PKG<
Ch. 21 - Prob. 1ECh. 21 - Prob. 2ECh. 21 - Prob. 3ECh. 21 - Prob. 4ECh. 21 - Prob. 5ECh. 21 - Prob. 6ECh. 21 - Prob. 7ECh. 21 - What are the main classifications of hydrocarbons?...Ch. 21 - Prob. 9ECh. 21 - What are structural isomers? How do the properties...
Ch. 21 - Prob. 11ECh. 21 - Define each term related to optical isomerism:...Ch. 21 - Prob. 13ECh. 21 - Prob. 14ECh. 21 - Explain geometric isomerism in alkenes. How do the...Ch. 21 - Prob. 16ECh. 21 - Prob. 17ECh. 21 - Prob. 18ECh. 21 - Prob. 19ECh. 21 - Prob. 20ECh. 21 - Prob. 21ECh. 21 - Prob. 22ECh. 21 - Prob. 23ECh. 21 - Prob. 24ECh. 21 - Prob. 25ECh. 21 - Prob. 26ECh. 21 - Prob. 27ECh. 21 - Prob. 28ECh. 21 - Prob. 29ECh. 21 - Prob. 30ECh. 21 - Prob. 31ECh. 21 - Prob. 32ECh. 21 - Prob. 33ECh. 21 - Prob. 34ECh. 21 - Prob. 35ECh. 21 - Prob. 36ECh. 21 - Prob. 37ECh. 21 - Prob. 38ECh. 21 - Prob. 39ECh. 21 - Prob. 40ECh. 21 - Name each alkane.Ch. 21 - Prob. 42ECh. 21 - Prob. 43ECh. 21 - Draw a structure for each alkane. a....Ch. 21 - Prob. 45ECh. 21 - Complete and balance each hydrocarbon combustion...Ch. 21 - Prob. 47ECh. 21 - Prob. 48ECh. 21 - Prob. 49ECh. 21 - Prob. 50ECh. 21 - Name each alkene.Ch. 21 - Prob. 52ECh. 21 - Prob. 53ECh. 21 - Name each alkyne.Ch. 21 - Draw the correct structure for each compound. a....Ch. 21 - Prob. 56ECh. 21 - Prob. 57ECh. 21 - Prob. 58ECh. 21 - Prob. 59ECh. 21 - Prob. 60ECh. 21 - Prob. 61ECh. 21 - Prob. 62ECh. 21 - Prob. 63ECh. 21 - Prob. 64ECh. 21 - Prob. 65ECh. 21 - Prob. 66ECh. 21 - Prob. 67ECh. 21 - Prob. 68ECh. 21 - Prob. 69ECh. 21 - Prob. 70ECh. 21 - Prob. 71ECh. 21 - Prob. 72ECh. 21 - Prob. 73ECh. 21 - Prob. 74ECh. 21 - Prob. 75ECh. 21 - Prob. 76ECh. 21 - Prob. 77ECh. 21 - Prob. 78ECh. 21 - Prob. 79ECh. 21 - Prob. 80ECh. 21 - Prob. 81ECh. 21 - Prob. 82ECh. 21 - Prob. 83ECh. 21 - Prob. 84ECh. 21 - Prob. 85ECh. 21 - Prob. 86ECh. 21 - Prob. 87ECh. 21 - Prob. 88ECh. 21 - Identify each organic compound as an alkane,...Ch. 21 - Identify each organic compound as an alkane,...Ch. 21 - Prob. 91ECh. 21 - Prob. 92ECh. 21 - Prob. 93ECh. 21 - Determine whether each pair of structures are...Ch. 21 - Prob. 95ECh. 21 - Prob. 96ECh. 21 - Classify each organic reaction as combustion,...Ch. 21 - Prob. 98ECh. 21 - Prob. 99ECh. 21 - Prob. 100ECh. 21 - Prob. 101ECh. 21 - Prob. 102ECh. 21 - Prob. 103ECh. 21 - Prob. 104ECh. 21 - Prob. 105ECh. 21 - Prob. 106ECh. 21 - Prob. 107ECh. 21 - Prob. 108ECh. 21 - Prob. 109ECh. 21 - Prob. 110ECh. 21 - Prob. 111ECh. 21 - Prob. 112ECh. 21 - Prob. 113ECh. 21 - Prob. 114ECh. 21 - Draw the structure and name a compound with the...Ch. 21 - Prob. 116ECh. 21 - Prob. 117ECh. 21 - Prob. 118ECh. 21 - Prob. 119ECh. 21 - Prob. 120ECh. 21 - Prob. 121ECh. 21 - Prob. 1SAQCh. 21 - Prob. 2SAQCh. 21 - Prob. 3SAQCh. 21 - Prob. 4SAQCh. 21 - Prob. 5SAQCh. 21 - Prob. 6SAQCh. 21 - Prob. 7SAQCh. 21 - Prob. 8SAQCh. 21 - Prob. 9SAQCh. 21 - Prob. 10SAQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Indicate the functions that salt bridges have in batteries.arrow_forwardIn the battery:Pt | H2 (g) | H+ (aq) | Fe2+ (aq) | FeIndicate the cathode and anode.arrow_forwardWrite the equations that occur when the electrode Pb (s) | PbI2 (s) | KI (ac) in a galvanic cell. a) It functions as a positive electrode b) It functions as a negative electrode c) What is the ion with respect to which this electrode is reversible?arrow_forward
- State the formula to find the electromotive force of a battery as a function of the potential of the anode and the cathode.arrow_forwardWhy are normal electrode potentials also called relative electrode potentials?arrow_forwardEasily differentiate between electrochemical potential and Galvani potential.arrow_forward
- Construct a molecular orbital diagram for carbon monoxide. Identify the relevant point group,include all of the appropriate symmetry labels and pictures, and fill in the electrons. Make sure toaccount for the difference in electronegativity between C and O. Hint: CO is substantiallyisoelectronic to N2. (PLEASE DRAW THE ENTIRE MO DIAGRAM!!!)arrow_forwardplease help with hwarrow_forwardhelp me solve this hwarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY