
Concept explainers
(a)
Interpretation:
The product formed when cholesterol reacts with hydrogen in presence of palladium-carbon has to be drawn.
(a)
Explanation of Solution
Cholesterol with hydrogen:
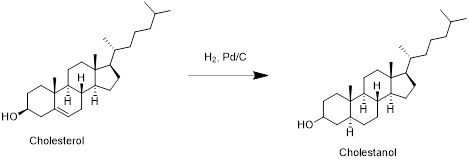
Cholesterol reacts with hydrogen in presence of palladium-carbon to reduce double bond to single bond and forms cholestanol. Therefore, the product obtained is cholestanol which is shown in the above scheme.
(b)
Interpretation:
The product formed when cholesterol reacts with acetyl chloride has to be drawn
(b)
Explanation of Solution
Cholesterol with acetyl chloride:
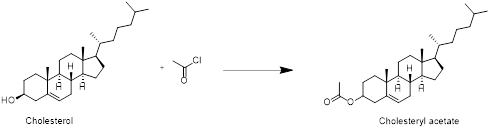
Cholesterol reacts with acetyl chloride in which acetylation reaction take place at hydroxyl group and forms cholesteryl acetate. Therefore, the product obtained is cholesteryl acetate which is shown in the above scheme.
(c)
Interpretation:
The product formed when cholesterol reacts with sulfuric acid followed by heating has to be drawn.
(c)
Explanation of Solution
Cholesterol with sulfuric acid:
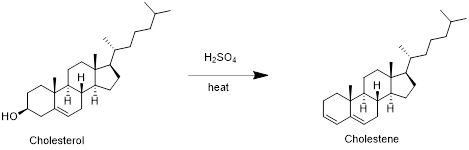
Cholesterol reacts with sulfuric acid in which removal of water molecule take place and forms cholestene. Therefore, the product obtained is cholestene which is shown in the above scheme.
(d)
Interpretation:
The product formed when cholesterol reacts with water in presence of acid has to be drawn.
(d)
Explanation of Solution
Cholesterol with water:
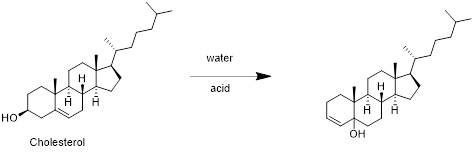
Cholesterol reacts with water in presence of water in which removal of water molecule take place and forms desired product. Therefore, the product obtained is is shown in the above scheme.
(e)
Interpretation:
The product formed when cholesterol reacts with peroxy acid has to be drawn
(e)
Explanation of Solution
Cholesterol with peroxy acid:
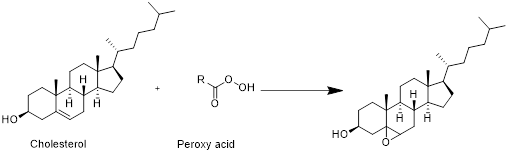
Cholesterol reacts with peroxy acid in presence of water in which reaction take place at double bond and forms
Want to see more full solutions like this?
Chapter 20 Solutions
EBK ESSENTIAL ORGANIC CHEMISTRY
- Draw the product of the E2 reaction shown below. Include the correct stereochemistry. Ignore any inorganic byproducts. + Br CH3 Q Strong Base Drawing Atoms, Bonds and Rings Charges Undo Reset H "Br H N Br. Remove Done .N. Drag To Panarrow_forwardCurved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and follow the curved arrows to draw the product of this elementary step in an elimination mechanism. Include all lone pairs and charges as appropriate. Ignore stereochemistry. Ignore byproducts. + Br: .. 8 0.01 M NaOH heat Drawing Q Atoms, Bonds and Rings Charges and Lone Pairs Draw or tap a new bond to see suggestions. Undo Reset Remove Done + Drag To Panarrow_forward+ Draw the product of the E2 reaction shown below. Include the correct stereochemistry. Ignore any inorganic byproducts. Ph CH2CH3 H H3C H Br DBN [૪] Drawing Atoms, Bonds and Rings H | OH Charges ―00 H. C | Undo Reset Br I Remove Done Drag To Pan +arrow_forward
- Reaction A Now the production A ΠIn the product of reaction i 12 Dear the product of actionarrow_forwardMacmillan Learnin When an unknown amine reacts with an unknown acid chloride, an amide with a molecular mass of 163 g/mol (M* = 163 m/z) is formed. In the infrared spectrum, important absorptions appear at 1661, 750 and 690 cm-1. The 13C NMR and DEPT spectra are provided. Draw the structure of the product as the resonance contributor lacking any formal charges. 13C NMR DEPT 90 200 160 120 80 40 0 200 160 120 80 DEPT 135 200 160 120 80 40 0 Draw the unknown amide. 40 40 0arrow_forwardDraw the major product karmed when I reach with the epoxide. Use walge dah bonds, including hydrogen al alcach genic center, to show the chemistry of the product Beeldraw any hydrogen akams on coxygen where applicablearrow_forward
- Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. H I Select to Add Arrows + H H 'H Q H2O H2O CI:O .H H H H I Select to Add Arrows I : C H2O H H H Select to Add Arrows 'Harrow_forward+ Draw an alkyl halide that produces ONLY the following alkene in an E2 elimination. Ignore any inorganic byproducts. Drawing Strong Base Q Atoms, Bonds and Rings Charges HO Br H2N Undo Reset Remove Done Drag To Panarrow_forwardFor the dehydrohalogenation (E2) reaction shown, draw the major organic product. Хок Br tert-butanol heat Select Drew Templates More Erase CH QQQarrow_forward
- Macmillan Learning Draw the major, neutral organic product for each substitution reaction. For this question, assume that each substitution reaction goes to completion. Disregard elimination. Reaction A. CI H₂O Select Draw Templates More Erase C Harrow_forwardMacmillan Learning Reaction B: CI HO_ 곳으 / Select Draw Templates More с € H D Erasearrow_forwardWhen 2-bromo-93-dimethylbutane is heated with sodium methoxide, one majors.. në la formed. 4th attempt Part 1 (0.5 point) t Ji See Periodic Table See Hint Draw the major alkene product and all other byproducts. Be sure to include lone-pair electrons and charges. Part 2 (0.5 point) What type of mechanism is occuring? Choose one: AS1 3rd attempt X H 41 See Hint Part 1 (0.5 point) Feedback See Periodic Table See Hintarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

