
FUND.OF PHYSICS(LL)-PRINT..W/ACCESS
12th Edition
ISBN: 9781119773504
Author: Halliday
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 19, Problem 8Q
The dot in Fig. 19-18c represents the initial state of a gas, and the adiabat through the dot divides the p-V diagram into regions 1 and 2. For the following processes, determine whether the corresponding heat Q is positive, negative, or zero: (a) the gas moves up along the adiabat, (b) it moves down along the adiabat, (c) it moves to anywhere in region 1, and (d) it moves to anywhere in region 2.
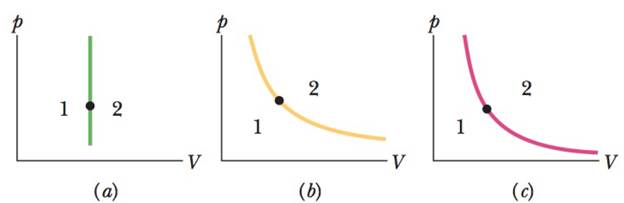
Figure 19-18 Questions 4, 6, and 8
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
I need help with how to solve this answer, thank you !
I need help with how to solve this answer, thank you !
I need help with how to solve this answer, thank you !
Chapter 19 Solutions
FUND.OF PHYSICS(LL)-PRINT..W/ACCESS
Ch. 19 - For four situations for an ideal gas, the table...Ch. 19 - For a temperature increase of T1, a certain amount...Ch. 19 - a Rank the four paths of Fig. 19-16 according to...Ch. 19 - The dot in Fig. 19-18c represents the initial...Ch. 19 - Prob. 9QCh. 19 - Does the temperature of an ideal gas increase,...Ch. 19 - Prob. 1PCh. 19 - Gold has a molar mass of 197 g/mol. a How many...Ch. 19 - SSM Oxygen gas having a volume of 1000 cm3 at...Ch. 19 - A quantity of ideal gas at: 10.0C and 100 kPa...
Ch. 19 - The best laboratory vacuum has a pressure of about...Ch. 19 - Water bottle in a hot car. In the American...Ch. 19 - Suppose 1.80 mol of an ideal gas is taken from a...Ch. 19 - Compute a the number of moles and b the number of...Ch. 19 - An automobile tire has a volume of 1.64 102 m3...Ch. 19 - A container encloses 2 mol of an ideal gas that...Ch. 19 - SSM ILW WWW Air that initially occupies 0.140 m3...Ch. 19 - GO Submarine rescue. When the U.S. submarine...Ch. 19 - In the temperature range 310 K to 330 K, the...Ch. 19 - An air bubble of volume 20 cm3 is at the bottom of...Ch. 19 - The temperature and pressure in the Suns...Ch. 19 - Calculate the rms speed of helium atoms at 1000 K....Ch. 19 - Find the rms speed of argon atoms at 313 K. See...Ch. 19 - A beam of hydrogen molecules H2 is directed toward...Ch. 19 - Prob. 25PCh. 19 - Prob. 26PCh. 19 - Water standing in the open at 32.0C evaporates...Ch. 19 - At what frequency would the wavelength of sound in...Ch. 19 - Prob. 30PCh. 19 - In a certain particle accelerator, protons travel...Ch. 19 - Prob. 32PCh. 19 - Prob. 33PCh. 19 - Prob. 34PCh. 19 - Prob. 35PCh. 19 - The most probable speed of the molecules in a gas...Ch. 19 - Two containers are at the same temperature. The...Ch. 19 - What is the internal energy of 1.0 mol of an ideal...Ch. 19 - Prob. 43PCh. 19 - ILW The mass of a gas molecule can be computed...Ch. 19 - Under constant pressure, the temperature of 2.00...Ch. 19 - The temperature of 2.00 mol of an ideal monatomic...Ch. 19 - GO When 20.9 J was added as heat to a particular...Ch. 19 - SSM A container holds a mixture of three...Ch. 19 - We give 70 J as heat to a diatomic gas, which then...Ch. 19 - Prob. 51PCh. 19 - SSM WWW Suppose 4.00 mol of an ideal diatomic gas...Ch. 19 - We know that for an adiabatic process pV = a...Ch. 19 - A certain gas occupies a volume of 4.3 L at a...Ch. 19 - Suppose 1.00 L of a gas with = 1.30, initially at...Ch. 19 - The volume of an ideal gas is adiabatically...Ch. 19 - GO Opening champagne. In a bottle of champagne,...Ch. 19 - GO Adiabatic wind. The normal airflow over the...Ch. 19 - GO A gas is to be expanded from initial state i to...Ch. 19 - GO An ideal diatomic gas, with rotation but no...Ch. 19 - Calculate the work done by an external agent...Ch. 19 - An ideal gas undergoes an adiabatic compression...Ch. 19 - Prob. 66PCh. 19 - An ideal monatomic gas initially has a temperature...Ch. 19 - Prob. 68PCh. 19 - SSM The envelope and basket of a hot-air balloon...Ch. 19 - An ideal gas, at initial temperature T1 and...Ch. 19 - Prob. 71PCh. 19 - Prob. 73PCh. 19 - Prob. 74PCh. 19 - The temperature of 3.00 mol of a gas with CV =...Ch. 19 - During a compression at a constant pressure of 250...Ch. 19 - Prob. 78PCh. 19 - SSM An ideal gas undergoes isothermal compression...Ch. 19 - Oxygen O2 gas at 273 K and 1.0 atm is confined to...Ch. 19 - An ideal pas is taken through a complete cycle in...Ch. 19 - Prob. 82PCh. 19 - SSM A sample of ideal gas expands from an initial...Ch. 19 - An ideal gas with 3.00 mol is initially in state 1...Ch. 19 - Prob. 85PCh. 19 - Prob. 86PCh. 19 - Prob. 87PCh. 19 - Prob. 88PCh. 19 - Prob. 89PCh. 19 - Prob. 90PCh. 19 - Prob. 91P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Describe an example of bioconversion. What metabolic processes can result in fuels?
Microbiology: An Introduction
Which of the following statements about the general functions of the nervous system is false?
The three primary...
Human Anatomy & Physiology (2nd Edition)
Johnny was vigorously exercising the only joints in the skull that are freely movable. What would you guess he ...
Anatomy & Physiology (6th Edition)
As genetic testing becomes widespread, medical records will contain the results of such testing. Who should hav...
Concepts of Genetics (12th Edition)
l. Suppose you have the uniformly charged cube in FIGURE Q24.1. Can you use symmetry alone to deduce the shape ...
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
8. A ball is thrown straight up into the air. At each of the following instants, is the ball's acceleration a? ...
College Physics: A Strategic Approach (3rd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Is work function of a metals surface related to surface energy and surface tension? What is the need to the work function component in the math of tension of metal surfaces that cannot be provided by existing equations of surface energy and surface tension? What are the key differences in each parameter and variables that allow for a differentiation of each function? What has a more significant meaning work function, surface tension or surface energy? Are there real differences and meaning? Please clarify and if possible provide examples . Does surface tension dependant on thickness of a metal or type of metal surface all having the same thickness? Clearly temperature has a profound change on surface tension what other variables besides temperature are key to surface tension. What if any is there a connection between crystal structure of the element and surface energy and tension? This is NOT a Assignment Question!!!arrow_forwardThe cylindrical beam of a 12.7-mW laser is 0.920 cm in diameter. What is the rms value of the electric field? V/marrow_forwardConsider a rubber rod that has been rubbed with fur to give the rod a net negative charge, and a glass rod that has been rubbed with silk to give it a net positive charge. After being charged by contact by the fur and silk...? a. Both rods have less mass b. the rubber rod has more mass and the glass rod has less mass c. both rods have more mass d. the masses of both rods are unchanged e. the rubber rod has less mass and the glass rod has mroe massarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning


Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers with Modern ...
Physics
ISBN:9781337553292
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:9781337553278
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning
Thermodynamics: Crash Course Physics #23; Author: Crash Course;https://www.youtube.com/watch?v=4i1MUWJoI0U;License: Standard YouTube License, CC-BY