
Chemistry: Structure and Properties (2nd Edition)
2nd Edition
ISBN: 9780134293936
Author: Nivaldo J. Tro
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 18, Problem 84E
Interpretation Introduction
(b)
To determine:
The sign of ∆Ssys for process of dew forming.
Interpretation Introduction
(c)
To determine:
The sign of ∆Ssys for the process shown in figure:
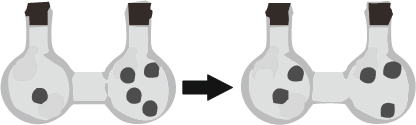
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the product formed when the following pair of compounds is treated with NaOEt in ethanol.
+
i
CN
I need help with the following
Explain the reasons for the color changes that occur when the gel and the solution examined dry.
Chapter 18 Solutions
Chemistry: Structure and Properties (2nd Edition)
Ch. 18 - What is the first law of thermodynamics, and how...Ch. 18 - What is nature’s heat tax, and how does it relate...Ch. 18 - What is a perpetual motion machine? Can such a...Ch. 18 - Is it more efficient to heat your home with a...Ch. 18 - What is a spontaneous process? Provide an example.Ch. 18 - Explain the difference between the spontaneity of...Ch. 18 - What is the precise definition of entropy? What is...Ch. 18 - Why does the entropy of a gas increase when it...Ch. 18 - Explain the difference between macrostates and...Ch. 18 - Based on its fundamental definition, explain why...
Ch. 18 - State the second law of thermodynamics. How does...Ch. 18 - What happens to the entropy of a sample of matter...Ch. 18 - Explain why water spontaneously freezes to form...Ch. 18 - Why do exothermic processes tend to be spontaneous...Ch. 18 - What is the significance of the change in Gibbs...Ch. 18 - Predict the spontaneity of a reaction (and the...Ch. 18 - State the third law of thermodynamics and explain...Ch. 18 - Why is the standard entropy of a substance in the...Ch. 18 - How does the standard entropy of a substance...Ch. 18 - How can you calculate the standard entropy change...Ch. 18 - Describe the three different methods to calculate...Ch. 18 - Why is free energy “free”?Ch. 18 - Explain the difference between G and G .Ch. 18 - Why does water spilled on the floor evaporate even...Ch. 18 - How do you calculate the change ¡n free energy for...Ch. 18 - How does the value of G for a reaction relate to...Ch. 18 - Prob. 27ECh. 18 - Prob. 28ECh. 18 - Prob. 29ECh. 18 - Prob. 30ECh. 18 - Calculate the change in entropy that occurs in the...Ch. 18 - Prob. 32ECh. 18 - Calculate the change ¡n entropy that occurs in the...Ch. 18 - Prob. 34ECh. 18 - Without doing any calculations, determine the sign...Ch. 18 - Prob. 36ECh. 18 - Without doing any calculations, determine the sign...Ch. 18 - Prob. 38ECh. 18 - Calculate Ssurr at the indicated temperature for...Ch. 18 - Prob. 40ECh. 18 - Given the values of Hrxn , Srxn and T, determine...Ch. 18 - Prob. 42ECh. 18 - Prob. 43ECh. 18 - Prob. 44ECh. 18 - Calculate the free energy change for the reaction...Ch. 18 - Prob. 46ECh. 18 - Prob. 47ECh. 18 - Predict the conditions (high temperature, low...Ch. 18 - How does the molar entropy of a substance change...Ch. 18 - What is the molar entropy of a pure crystal at 0...Ch. 18 - For each pair of substances, choose the one that...Ch. 18 - For each pair of substances, choose the one that...Ch. 18 - Rank each set of substances in order of increasing...Ch. 18 - Prob. 54ECh. 18 - Use data from Appendix IIB to calculate Srxn for...Ch. 18 - Use data from Appendix IIB to calculate Srxn for...Ch. 18 - Find S for the formation of CH2Cl2(g) from its...Ch. 18 - Prob. 58ECh. 18 - Methanol burns in oxygen to form carbon dioxide...Ch. 18 - In photosynthesis, plants form glucose (C6H12O6)...Ch. 18 - For each reaction, calculate Hrxn , Srxn and Grxn...Ch. 18 - For each reaction calculate Hrxn , Srxn and Grxn...Ch. 18 - Use standard free energies of formation to...Ch. 18 - Use standard free energies of formation to...Ch. 18 - Consider the reaction: 2NO(g)+O2(g)2NO2(g)...Ch. 18 - Prob. 66ECh. 18 - Determine G for the reaction:...Ch. 18 - Prob. 68ECh. 18 - Consider the sublimation of iodine at 25.0°C:...Ch. 18 - Consider the evaporation of methanol at 25.0°C....Ch. 18 - Consider the reaction: CH3OH(g)CO(g)+2H2(g)...Ch. 18 - Consider the reaction: CO2(g)+CCl4(g)2COCl2(g)...Ch. 18 - Use data from Appendix IIB to calculate the...Ch. 18 - Prob. 74ECh. 18 - Prob. 75ECh. 18 - Prob. 76ECh. 18 - Prob. 77ECh. 18 - Prob. 78ECh. 18 - Consider the reaction: H2(g)+I2(g)2HI(g) The...Ch. 18 - Consider the reaction: 2N0(g) — O(g) 2N02(g) The...Ch. 18 - The change in enthalpy (Hrxn) for a reaction is...Ch. 18 - Prob. 82ECh. 18 - Prob. 83ECh. 18 - Prob. 84ECh. 18 - Our atmosphere is composed primarily of nitrogen...Ch. 18 - Prob. 86ECh. 18 - Ethene (C2H4) can be halogenated by the reaction:...Ch. 18 - H2 reacts with the halogens (X2) according to the...Ch. 18 - Consider this reaction occurring at 298 K:...Ch. 18 - Consider this reaction occurring at 298 K:...Ch. 18 - Prob. 91ECh. 18 - Prob. 92ECh. 18 - These reactions are important in catalytic...Ch. 18 - Prob. 94ECh. 18 - All the oxides of nitrogen have positive values of...Ch. 18 - Prob. 96ECh. 18 - Consider the reaction X2(g)2X(g) . When a vessel...Ch. 18 - Prob. 98ECh. 18 - Indicate and explain the sign of Suniv for each...Ch. 18 - The Haber process is very important for...Ch. 18 - A metal salt with the formula MCl2 crystallizes...Ch. 18 - The solubility of AgCI(s) in water at 25°C is...Ch. 18 - Review the subsection in this chapter entitled...Ch. 18 - Calculate the entropy of each state and rank the...Ch. 18 - Suppose we redefine the standard state as P=2atm ....Ch. 18 - The G for the freezing of H2O(l) at 10°C is 210...Ch. 18 - Consider the reaction that occurs during the Haber...Ch. 18 - The salt ammonium nitrate can follow three modes...Ch. 18 - Given the tabulated data, calculate Svap for each...Ch. 18 - Prob. 110ECh. 18 - Prob. 111ECh. 18 - Consider the changes in the distribution of nine...Ch. 18 - Prob. 113ECh. 18 - Prob. 114ECh. 18 - Prob. 115ECh. 18 - The reaction A(g)B(g) has an equilibrium constant...Ch. 18 - Prob. 117ECh. 18 - Prob. 118ECh. 18 - Prob. 119ECh. 18 - Have each group member look up Hf and S for one...Ch. 18 - Calculate G at 25°C for the reaction in the...Ch. 18 - Prob. 122ECh. 18 - Which reaction Is most likely to have a positive...Ch. 18 - Prob. 2SAQCh. 18 - Arrange the gases—F2, Ar, and CH3F—in order of...Ch. 18 - Prob. 4SAQCh. 18 - Prob. 5SAQCh. 18 - For a certain reaction Hrxn=255kJ and Srxn=211J/K...Ch. 18 - Prob. 7SAQCh. 18 - s8. Use standard free energies of formation to...Ch. 18 - Prob. 9SAQCh. 18 - For the following reaction, Grxn=9.4kJ at 25 °C....Ch. 18 - Prob. 11SAQCh. 18 - Prob. 12SAQCh. 18 - Prob. 13SAQCh. 18 - Prob. 14SAQCh. 18 - Prob. 15SAQCh. 18 - Prob. 16SAQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- For Raman spectroscopy/imaging, which statement is not true regarding its disadvantages? a) Limited spatial resolution. b) Short integration time. c) A one-dimensional technique. d) Weak signal, only 1 in 108 incident photons is Raman scattered. e) Fluorescence interference.arrow_forwardUsing a cell of known pathlength b = 1.25115 x 10-3 cm, a water absorption spectrum was measured. The band at 1645 cm-1, assigned to the O-H bending, showed an absorbance, A, of 1.40. a) Assuming that water density is 1.00 g/mL, calculate the water molar concentration c (hint: M= mole/L) b) Calculate the molar absorptivity, a, of the 1645 cm-1 band c) The transmitted light, I, can be written as I= Ioexp(-xb), where x is the absorption coefficient (sometimes designated as alpha), Io is the input light, and b is the cell pathlength. Prove that x= (ln10)*x*c. (Please provide a full derivation of the equation for x from the equation for I). d) Calculate x for the 1645 cm-1 bandarrow_forwardI need help with the follloaingarrow_forward
- For a CARS experiment on a Raman band 918 cm-1, if omega1= 1280 nm, calculate the omega2 in wavelength (nm) and the CARS output in wavelength (nm).arrow_forwardI need help with the following questionarrow_forwardFor CARS, which statement is not true regarding its advantages? a) Contrast signal based on vibrational characteristics, no need for fluorescent tagging. b) Stronger signals than spontaneous Raman. c) Suffers from fluorescence interference, because CARS signal is at high frequency. d) Faster, more efficient imaging for real-time analysis. e) Higher resolution than spontaneous Raman microscopy.arrow_forward
- Draw the major product of the Claisen condensation reaction between two molecules of this ester. Ignore inorganic byproducts. Incorrect, 5 attempts remaining 1. NaOCH3/CH3OH 2. Acidic workup Select to Draw O Incorrect, 5 attempts remaining The total number of carbons in the parent chain is incorrect. Review the reaction conditions including starting materials and/or intermediate structures and recount the number of carbon atoms in the parent chain of your structure. OKarrow_forwardUsing a cell of known pathlength b = 1.25115 x 10-3 cm, a water absorption spectrum was measured. The band at 1645 cm-1, assigned to the O-H bending, showed an absorbance, A, of 1.40. a) Assuming that water density is 1.00 g/mL, calculate the water molar concentration c (hint: M= mole/L) b) Calculate the molar absorptivity, a, of the 1645 cm-1 band c) The transmitted light, I, can be written as I= Ioexp(-xb), where x is the absorption coefficient (sometimes designated as alpha), Io is the input light, and b is the cell pathlength. Prove that x= (ln10)*x*c d) Calculate x for the 1645 cm-1 bandarrow_forwardConvert 1.38 eV into wavelength (nm) and wavenumber (cm-1) (c = 2.998 x 108 m/s; h = 6.626 x 10-34 J*s).arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY