
Concept explainers
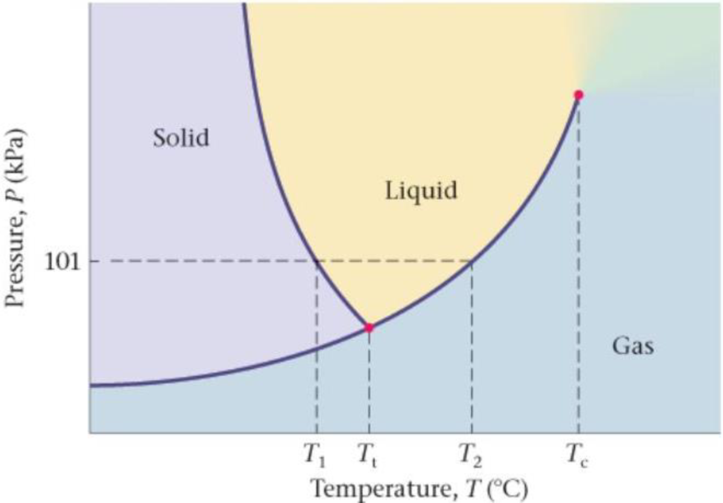
Phase Diagram for Water The phase diagram for water is shown in Figure 17-37. (a) What is the temperature T1 on the phase diagram? (b) What is the temperature T2 on the phase diagram? (c) What happens to the melting/freezing temperature of water if atmospheric pressure is decreased? Justify your answer by referring to the phase diagram (d) What happens to the boiling/condensation temperature of water if atmospheric pressure is increased? Justify your answer by referring to the phase diagram.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Physics (5th Edition)
Additional Science Textbook Solutions
Physics for Scientists and Engineers with Modern Physics
Essential University Physics (3rd Edition)
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Tutorials in Introductory Physics
University Physics (14th Edition)
Introduction to Electrodynamics
- I Review Quantity of heat: Q = cmAT Part A Find what heat in calories (cal) is required to increase the temperature of 60 g water from 0° C to 65 °C. The specific heat capacity of water is 1 cal/g.° C. Express your answer to two significant figures and include the appropriate units. HA Q = Value Units Submit Request Answerarrow_forwardPlease answer the 3 questions. Use the table below the solve the ff thermal expansion problems: An aluminum flagpole is 33 m high. By how much does its length increase as the temperature increases by 15 °C? At 20°C, a brass cube has edge length 30 cm. What is the increase in the surface area when it is heated from 20°C to 75°C? 3. What is the volume of a lead ball at 30.0°C if the ball’s volume at 60.0°C is 50.00 cm3?arrow_forward3 of 4 A stainless-steel-bottomed kettle, its bottom 25 cm in diameter and 1.6 mm thick, sits on a burner. The kettle holds boiling water, and energy flows into the water from the kettle bottom at 800 W. Review I Constants I Periodic Tabl Part A What is the temperature of the bottom surface of the kettle? Thermal conductivity of stainless steel is 14 W/(m K). Express your answer using four significant figures. VO AEO ? T = 102.65 °C Submit Previous Answers Request Answer X Incorrect; Try Again; 5 attempts remaining Review your calculations and make sure you round to 4 significant figures in the last step. Ne Provide Feedbackarrow_forward
- Insulating Material A Part A: 960 Btu/hr heat is conducted through a section of insulating material shown in Figure that measures 9.5 ft² in cross- sectional area. The k thickness is 0.725 inch. ΔΧ The thermal conductivity of the material is 0.19 W/m K. Calculate the, a) area of the sample in square meter (m²) : b) thickness of the specimen in meter (m): c) temperature difference in Kelvin :arrow_forwardConstants Periodic Table Assume all temperatures to be exact, and neglect significant figures for small changes in dimension. On a warm day (92 °F), an air-filled balloon occupies a volume of 0.300 m³ and has a pressure of 24.0 lb/in?. Part A If the balloon is cooled to 32 °F in a refrigerator while its pressure is reduced to 14.7 lb/in?, what is the volume of the air in the container? (Assume that the air behaves as an ideal gas.) nνα ΑΣφ V = 0.170 m3 Submit Previous Answers Request Answer X Incorrect; Try Again; 6 attempts remainingarrow_forwardNumber 6 & 7arrow_forward
- Please solve this on a sheet of paper.arrow_forwardConstants The 1.30-kg head of a hammer has a speed of 8.5 m/s just before it strikes a nail and is brought to rest (Figure 1). Part A Estimate the temperature rise of a 12-g iron nail generated by 6.0 such hammer blows done in quick succession. Assume the nail absorbs all the energy. The specific heat of iron is 450 J/kg C°. Express your answer using two significant figures. Figurearrow_forwardPlease explainarrow_forward
 College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning





