
(a)
Interpretation:
The acceptable name for the following amide should be determined:
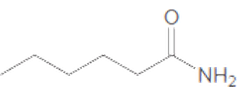
Concept Introduction:
An organic compound in which carboxy
The reaction which results in the formation of amide along with water on heating acids with
So, in order to give the IUPAC name to the amides, the rules for naming carboxylic acid is followed and -oic acid of the carboxylic acid is replaced by amide.
In order to give the name to the amide group, the following steps are followed:
- The parent (longest)
alkane chain is named as for carboxylic acids. - The -oic acid in the name is changed to -amide.
- The numbering of the chain is done in such a way that amide group and substituents gets the smaller number.
- N-alkyl is used to show each alkyl group bonded to -N atom in the name for secondary and tertiary amides.
For number of carbons atoms chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
In order to write the common name of the amides, the common of acids are written from which the amide has been formed by replacing -oic acid in name from -amide.
(b)
Interpretation:
The acceptable name for the following amide should be determined:
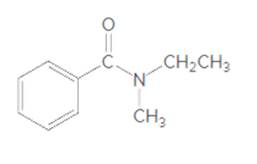
Concept Introduction:
An organic compound in which carboxy functional group that is -COOH is bonded to the carbon atom is said to be a carboxylic acid. The general formula for carboxylic acid is RCOOH or RCO2H. When -OH (hydroxyl group) of the carboxylic acid is replaced by nitrogen (-N) then it results in the formation of an amide.
The reaction which results in the formation of amide along with water on heating acids with amine or ammonia is said to be amidation.
So, in order to give the IUPAC name to the amides, the rules for naming carboxylic acid is followed and -oic acid of the carboxylic acid is replaced by amide.
In order to give the name to the amide group, the following steps are followed:
- The parent (longest) alkane chain is named as for carboxylic acids.
- The -oic acid in the name is changed to -amide.
- The numbering of the chain is done in such a way that amide group and substituents gets the smaller number.
- N-alkyl is used to show each alkyl group bonded to -N atom in the name for secondary and tertiary amides.
For number of carbons atoms chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
In order to write the common name of the amides, the common of acids are written from which the amide has been formed by replacing -oic acid in name from -amide.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Connect One Semester Access Card for General, Organic, & Biological Chemistry
- Steps and explanations pleasearrow_forwardUse diagram to answer the following: 1.Is the overall rxn endo- or exothermic. Explain briefly your answer____________________2. How many steps in this mechanism?_____________3. Which is the rate determining step? Explain briefly your answer____________________4. Identify (circle and label) the reactants,the products and intermediate (Is a Cation, Anion, or a Radical?) Please explain and provide full understanding.arrow_forwardDraw the entire mechanism and add Curved Arrows to show clearly how electrons areredistributed in the process. Please explain and provide steps clearly.arrow_forward
- Match the denticity to the ligand. Water monodentate ✓ C₂O2 bidentate H₂NCH₂NHCH2NH2 bidentate x EDTA hexadentate Question 12 Partially correct Mark 2 out of 2 Flag question Provide the required information for the coordination compound shown below: Na NC-Ag-CN] Number of ligands: 20 Coordination number: 2✔ Geometry: linear Oxidation state of transition metal ion: +3 x in 12 correct out of 2 question Provide the required information for the coordination compound shown below. Na NC-Ag-CN] Number of ligands: 20 Coordination number: 2 Geometry: linear 0 Oxidation state of transition metal ion: +3Xarrow_forwardCan you explain step by step behind what the synthetic strategy would be?arrow_forwardPlease explain step by step in detail the reasoning behind this problem/approach/and answer. thank you!arrow_forward
- 2. Predict the product(s) that forms and explain why it forms. Assume that any necessary catalytic acid is present. .OH HO H₂N OHarrow_forwardconsider the rate of the reaction below to be r. Whats the rate after each reaction? Br + NaCN CN + NaBr a. Double the concentration of alkyl bromide b. Halve the concentration of the electrophile & triple concentration of cyanide c. Halve the concentration of alkyl chloridearrow_forwardPredict the organic reactant that is involved in the reaction below, and draw the skeletal ("line") structures of the missing organic reactant. Please include all steps & drawings & explanations.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

