
Concept explainers
A hydrogen atom in the organic base pyridine, C5H5N, can be substituted by various atoms or groups to give XC5H4N, where X is an atom such as Cl or a group such as CH3. The following table gives Ka values for the conjugate acids of a variety of substituted pyridines.
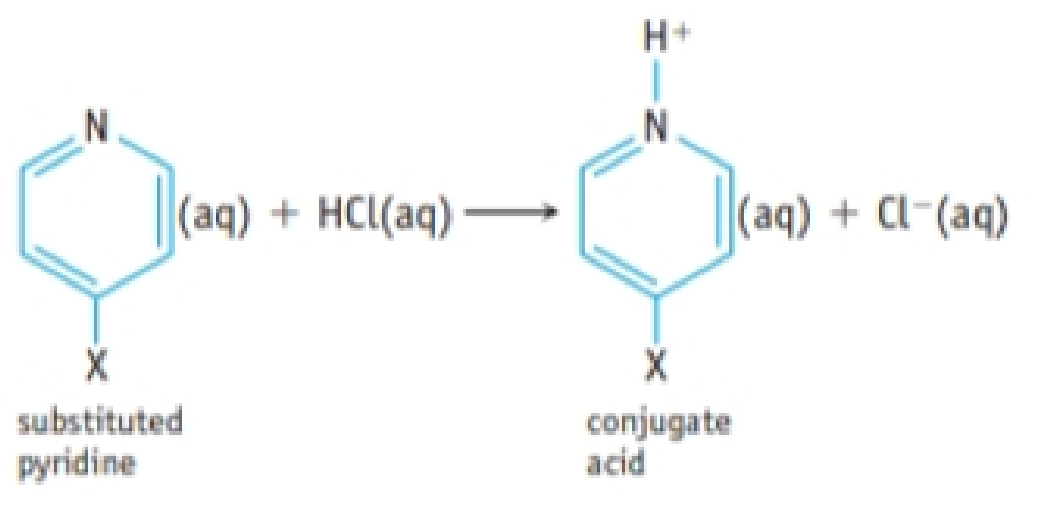
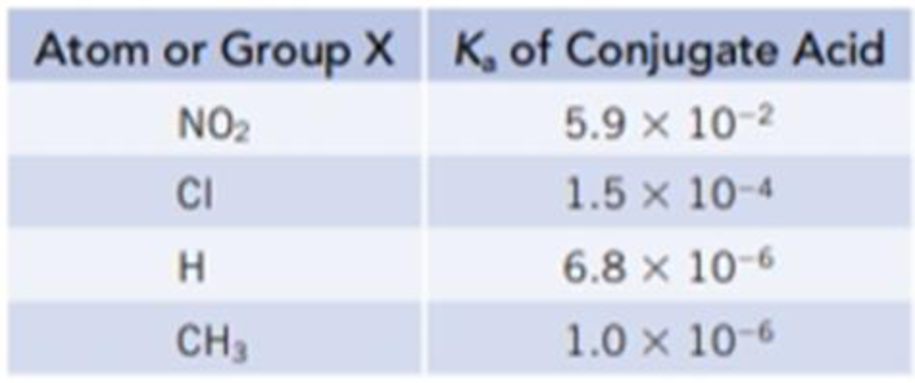
- (a) Suppose each conjugate acid is dissolved in sufficient water to give a 0.050 M solution. Which solution would have the highest pH? The lowest pH?
- (b) Which of the substituted pyridines is the strongest Brønsted base? Which is the weakest Brønsted base?
(a)
Interpretation:
The conjugate acid which have the highest
Concept Introduction:
The
Higher the value of
Answer to Problem 112IL
The solution containing
Explanation of Solution
An equilibrium constant
For Any acid HA,
The relative strength of an acid and base in water can be also expressed quantitatively with an equilibrium constant as follows:
An equilibrium constant
Given:
The
Initial Concentration of each solution is
Set up an ICE table for the reaction of
Substitute the values in equation (2) to calculate
Substitute the value of
The value of
Substitute the value of hydronium ion concentration in equation (1) to calculate the value of
Therefore, the value of
Similarly, Substitute the value of
The value of
Substitute the concentration of hydronium ion in equation (1) to calculate the value of
Therefore, the value of
Substitute the value of
The value of
Substitute the concentration of hydronium ion in equation (1) to calculate the value of
Therefore, the value of
Substitute the value of
The value of
Substitute the concentration of hydronium ion in equation (1) to calculate the value of
Therefore, the value of
The solution containing
(b)
Interpretation:
Strongest
Concept Introduction:
A conjugate acid-base pair contains two compounds that differ only by a hydrogen ion and a charge of
The stronger the acid, the weaker its conjugate base and vice-verca. That is, the larger the values of
Answer to Problem 112IL
The strongest bronsted base is
Explanation of Solution
The dissociation of
A small value of
The dissociation of
A large value of
The strongest bronsted base is
Want to see more full solutions like this?
Chapter 16 Solutions
Chemistry & Chemical Reactivity
- If the current voltage is n = 0.14 V. Indicate which of the 2 formulas must be applied a) = a T = i exp[(1 - p) F Fn Fn b) i==exp B RTarrow_forwardTopic: Photochemistry and Photophysics of Supramoleculesarrow_forwardTwo cations that exchange an electron in an interface, the exchange density is worth 1.39 mA/cm2 and the current density is worth 15 mA/cm2 at 25°C. If the overvoltage is 0.14 V, calculate the reaction rate and symmetry factor. Data: R = 8,314 J mol-1 k-1: F = 96500 Carrow_forward
- With the help of the Tafel line, it is estimated that the interchange density of the VO2+/VO2+ system on the carbon paper has a value of 3 mA cm-2. Calculate a) the current density if the voltage has a value of 1.6 mV and the temperature is 25°C. b) the beta value of the anódico process if the Tafel pendulum is 0.6 V at 25°C. Data: R = 8.314 JK-1mol-1, y F = 96485 C mol-1.arrow_forwardApply the NANSTE law to the MnO4- + 8H+ + 5e- ⇄ Mn2+ + 4H2Oarrow_forwardIn the Nernst Law, how much is RT / F?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning





