
(a)
Interpretation: For the given molecular formulae the structure should be predicted.
Concept Introduction:
1H NMR: A technique gives information to predict the Carbon and Hydrogen connectivity in the organic compounds.
HDI (Index of hydrogen deficiency): HDI values gives information on the level of unsaturation (means a double bond or a ring) on the structure of the compound.
(a)
Answer to Problem 32PP
Answer
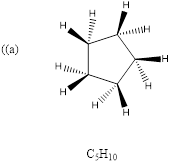
Explanation of Solution
To predict: the structure for the given molecular formula.
Calculate HDI Value for the given molecular formula and draw the structure.
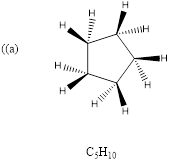
HDI = 1, compound has either 1 ring or one
Therefore, the compound predicted is the cyclopentane.
(b)
Interpretation: For the given molecular formulae the structure should be predicted.
Concept Introduction:
1H NMR: A technique gives information to predict the Carbon and Hydrogen connectivity in the organic compounds.
HDI (Index of hydrogen deficiency): HDI values gives information on the level of unsaturation (means a double bond or a ring) on the structure of the compound.
(b)
Answer to Problem 32PP
Answer
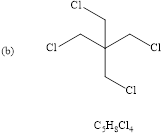
Explanation of Solution
The structure of given molecular formula.(
Calculate HDI value for the given molecular formula and draw the structure:

HDI = 0 , tells the compound is fully saturated (no double bond, triple bond or rings) and the compound is acyclic. Only one proton signal indicates all 4-
Therefore the predicted compound is 1, 3-dichoro-2, 2-dimethylchloropropane.
(c)
Interpretation: For the given molecular formulae the structure should be predicted.
Concept Introduction:
1H NMR: A technique gives information to predict the Carbon and Hydrogen connectivity in the organic compounds.
HDI (Index of hydrogen deficiency): HDI values gives information on the level of unsaturation (means a double bond or a ring) on the structure of the compound.
(c)
Answer to Problem 32PP
Answer
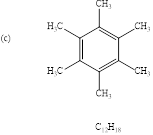
Explanation of Solution
To find: the structure of given molecular formula.
Calculate HDI value for the given molecular formula and draw the structure:
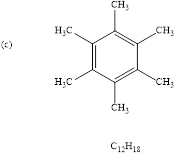
HDI = 4, shows the compound is
Want to see more full solutions like this?
Chapter 15 Solutions
ORGANIC CHEMISTRY-PRINT (LL)-W/WILEY
- Predict the major products of this organic reaction: HBr (1 equiv) cold ? Some important notes: • Draw the major product, or products, of this reaction in the drawing area below. • You can draw the products in any arrangement you like. • Pay careful attention to the reaction conditions, and only include the major products. • Be sure to use wedge and dash bonds when necessary, for example to distinguish between major products that are enantiomers. • Note that there is only 1 equivalent of HBr reactant, so you need not consider the case of multiple additions. dm Re Explanation Check ©2025 McGraw Hill LLC. All Rights Reserved. Termarrow_forwardb) Use curved arrows to show the reaction of the radical with hydrogen bromide. Br: Br H .. Answer Bankarrow_forwardIndicate the reaction products when CH3COCH2COOCH2COOC2H5 (ethyl acetoacetoacetate) reacts with 1º OH-/H2O and 2º H3O+arrow_forward
- Indicate whether the product of the reaction between Naphthalene and CrO3 in acetic acid at 25ºC is 1,4 naphthoquinone or phthalic anhydride.arrow_forwardIndicate the products of the reaction between CH3COCH2COOC2H5 and Na+-OC2H5.arrow_forwardPrimary, Secondary, and Tertiary Alcohols O-H O-H O-H R₁-C-H R₁-C-H R₁-C-R₁ H R₂ R₂ Primary Alcohol Secondary Alcohol ChemistryLearner.com R stands for Carbon group like ethyl methyl propyl Tertiary Alcohol If 1 carbon group with two H attached to alcoholic carbon, then primary If 2 carbon group and 1 H are attached to alcoholic carbon, then secondary IF 3 carbon group and no H attach to alcoholic carbon then tertiary. The bottom line Starting "Weak" oxidant material PCC, DMP, Swern, etc Primary alcohol Aldehyde OH Secondary alcohol Ketone OH "Strong" oxidant KMnO4, H₂CrO4 (or equivalent) OH Carboxylic acid 요 Ketone No reaction No reaction Tertiary alcohol 1. Is ethanol a primary, secondary, or tertiary alcohol? Write out the structures of ethanol and any oxidation products of ethanol. If there is more than one oxidation product, give the structure of each of the products. 2. Is 2-propanol a primary, secondary, or tertiary alcohol? Write out the structures of 2-propanol and any…arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





