
Concept explainers
(a)
Interpretation:
How UV-vis spectroscopy could be used to determine whether the given reaction has actually taken place or not is to be suggested with reason.
Concept introduction:
Compounds with lone pairs tend to have longer-wavelength absorptions than analogous compounds without lone pairs. The longest
Answer to Problem 15.36P
The given reaction will take place as suggested by change in the
Explanation of Solution
The given reaction is:
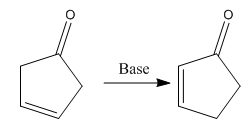
In the above reaction, both the reactant and product have two double bonds; in the product, both double bonds are in conjugation, but in the reactant, they are not.
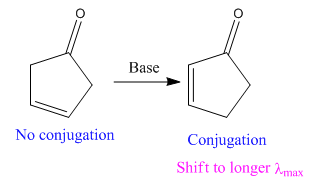
Thus, we can say that the given reaction has actually occurred.
Whether the given reaction has actually taken place is suggested on the basis of conjugation in the product and its effect on
(b)
Interpretation:
How UV-vis. Spectroscopy could be used to determine whether the given reaction has actually taken place or not is to be suggested with reason.
Concept introduction:
Compounds with lone pairs tend to have longer-wavelength absorptions than analogous compounds without lone pairs. The longest
Answer to Problem 15.36P
The given reaction does not occur as given, since the
Explanation of Solution
The given reaction is:
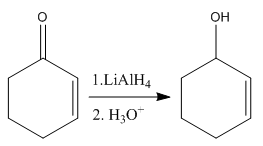
In the above reaction, the reactant has two double bonds in conjugation, and the product has only one double bond. Thus,
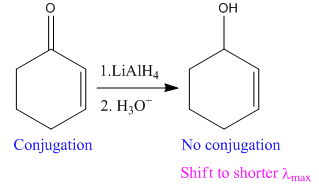
Thus, we can say that the given reaction does not take place.
Whether the given reaction has actually taken place is suggested on the basis of conjugation in the product and its effect on
(c)
Interpretation:
How UV-vis. Spectroscopy could be used to determine whether the given reaction has actually taken place or not is to be suggested with reason.
Concept introduction:
Compounds with lone pairs tend to have longer-wavelength absorptions than analogous compounds without lone pairs. The longest
Answer to Problem 15.36P
The given reaction not take place as given, since the
Explanation of Solution
The given reaction is:
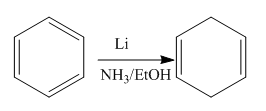
In the above reaction, the reactant has three double bonds in conjugation, and the product has two double bonds not in conjugation. The value of the longest-wavelength
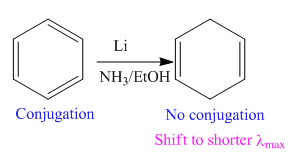
Thus,
Whether the given reaction actually took is suggested on the basis of conjugation in the product and its effect on
(d)
Interpretation:
How UV-vis. Spectroscopy could be used to determine whether the given reaction has actually taken place or not is to be suggested with reason.
Concept introduction:
Compounds with lone pairs tend to have longer-wavelength absorptions than analogous compounds without lone pairs. The longest
Answer to Problem 15.36P
The given reaction will take place as suggested by change in the
Explanation of Solution
The given reaction is:
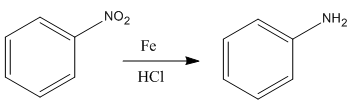
In the above reaction, both reactant and product have three triple bonds in conjugation, but the reactant has a lone pair. So, the reactant’s longest-wavelength absorption corresponds to a
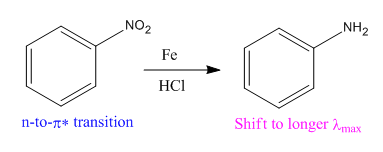
Therefore the
Whether the given reaction actually took is suggested on the basis of conjugation in the product and its effect on
(e)
Interpretation:
How UV-vis. Spectroscopy could be used to determine whether the given reaction actually took is to be suggested with reason.
Concept introduction:
Compounds with lone pairs tend to have longer-wavelength absorptions than analogous compounds without lone pairs. The longest
Answer to Problem 15.36P
The given reaction will take place as suggested by change in the
Explanation of Solution
The given reaction is:
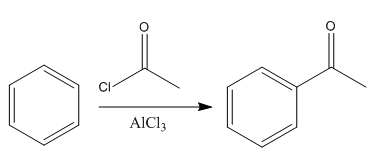
In the above reaction, the reactant has three conjugated bonds, and the product has four conjugated double bonds.
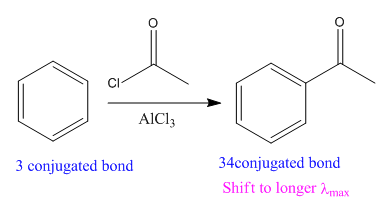
Thus, we can say that the given reaction is actually took place.
Whether the given reaction actually took is suggested on the basis of conjugation in the product and its effect on
Want to see more full solutions like this?
Chapter 15 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- : At a certain temperature, the equilibrium constant K for the following reaction is 1.58 × 10-12 N2(g) + O2(g) = 2 NO(g) Use this information to complete the following table. Suppose a 38. L reaction vessel is filled with 0.93 mol of N2 and 0.93 mol of O2. What can you say about the composition of the mixture in the vessel at equilibrium? There will be very little N2 and O2. There will be very little NO. What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. 2 NO(g) N2(9)+02(9) What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. 3 N2(9)+302(g) 6 NO(g) Neither of the above is true. K = ☐ K = ☐ ☐ X10 Х D ? 000 18 Ar Barrow_forwardwhen performing the reaction that involves 2 equivalents of 3-(diethylamino)-phenol and Phthalic anhydride with sulfuric acid and water react to form rhodamine b where the Phthalic anhydride cleaves in acid and how does Excessive Washing (w/ Base) & Subsequent Resonance Structure get affectedarrow_forward3. The strongest acid of the following compounds is ___.A. p-nitrophenol; B. m-nitrophenol; C. o-chlorophenol;D. p-methoxyphenol; E. o-methylphenol Please explain your steps and thought process. Thank you!arrow_forward
- Using the general properties of equilibrium constants At a certain temperature, the equilibrium constant K for the following reaction is 1.3 × 10 4: Cl2(g) + CHCl3(g) HCl(g) + CC₁(g) Use this information to complete the following table. Suppose a 16. L reaction vessel is filled with 1.6 mol of HCI and 1.6 mol of CCl4. What can you say about the composition of the mixture in the vessel at equilibrium? There will be very little Cl2 and CHCl3. ☐ x10 There will be very little HCI and CCl4. Neither of the above is true. What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. HCl(g)+CC14(g) 12 Cl2(9)+CHCl3(9) K = 0 ☐ What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. 2 Cl₂(9)+2CHCl3(9) 2 HCl(9)+2CC₁₁(9) K = ✓ 00. 18 Ararrow_forward10. The most important reason why Br- is a better nucleophile than Cl-is ___. A. polarizability; B. size; C. solvation; D. basicity; E. polarity. Please include all steps. Thanks!arrow_forwardPredicting the qualitative acid-base properties of salts Consider the following data on some weak acids and weak bases: base acid Ка K₁₁ name formula name formula nitrous acid HNO2 4.5×10 4 pyridine CHEN 1.7 × 10 9 4 hydrofluoric acid HF 6.8 × 10 methylamine CH3NH2 | 4.4 × 10¯ Use this data to rank the following solutions in order of increasing pH. In other words, select a '1' next to the solution that will have the lowest pH, a '2' next to the solution that will have the next lowest pH, and so on. solution 0.1 M NaNO2 0.1 M KF pH choose one v choose one v 0.1 M C5H5NHBr 0.1 M CH3NH3CI choose one v ✓ choose one 1 (lowest) 2 ☑ 3 4 (highest) 000 18 Ararrow_forward
- 4. The major product from treatment of 2-propanol with the Jonesreagent is ___.A. acetone; B. none of the other answers is correct C. propene; D.propanoic acid; E carbon dioxide. Please include all steps! Thank you!arrow_forward7. All of the following compounds that are at the same oxidation levelare ___.u. methyl epoxide, v. propyne, w. propanal, x. propene,y. 2,2-dihydroxypropane, z. isopropanol?A. u,v,w,y; B. u,v,w; C. v,w,y,z; D. v, z; E. x,y,z Please include all steps. Thank you!arrow_forward9. Which one of the following substituents is the worst leaving group inan SN2 reaction? A. -NH2; B. -OH; C. –F; D. NH3; E. H2O Please include all steps. Thanks!arrow_forward
- Using the general properties of equilibrium constants At a certain temperature, the equilibrium constant K for the following reaction is 2.5 × 105: CO(g) + H2O(g) CO2(g) + H2(g) Use this information to complete the following table. Suppose a 7.0 L reaction vessel is filled with 1.7 mol of CO and 1.7 mol of H2O. What can you say about the composition of the mixture in the vessel at equilibrium? What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. CO2(9)+H2(g) CO(g)+H₂O(g) What is the equilibrium constant for the following reaction? Be sure your answer has the correct number of significant digits. 3 CO(g)+3H2O(g) = 3 CO2(g)+3H2(g) There will be very little CO and H2O. x10 There will be very little CO2 and H2. 000 Neither of the above is true. K = ☐ K = ☐ 18 Ararrow_forward8. When ethane thiol is treated with hydrogen peroxide the product is___.A. ethane disulfide; B. diethyl sulfide; C. ethane sulfoxide; D. ethanesulfate; E. ethyl mercaptan. Please include all steps. Thanks!arrow_forward5. The major product of the three step reaction that takes place when 1-propanol is treated with strong acid is?A. dipropyl ether; B. propene; C. propanal; D. isopropyl propyl ether;E. 1-hexanol Please include all steps. Thank you!arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

