
Concept explainers
(a)
Interpretation:
The possible structures of the starting materials of the given Diels-Alder product are to be given.
Concept introduction:
In order to find the starting materials of the given compounds follows the rules are given below.
First, label the double bond carbons as
In order to complete the diene structure, eliminate the double bonds between
In order to complete the dienophile structure, add the double bonds between its carbons.
Also, dienophile is substituted with electron withdrawing groups.
Answer to Problem 15.20P
The possible structures of starting materials of given compound can be as follows.
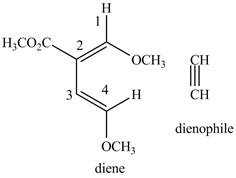
Explanation of Solution
First designate the double bond carbon atoms as
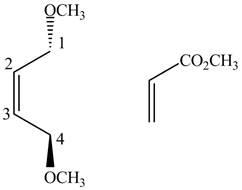
Figure 1
In order to complete the structure of diene, remove the double bonds present between carbons
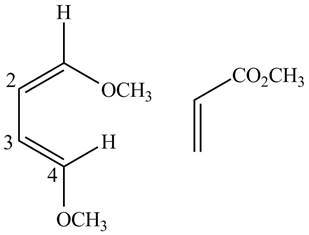
Figure 2
Finally, complete the dienophile structure by adding the double bonds between carbons.
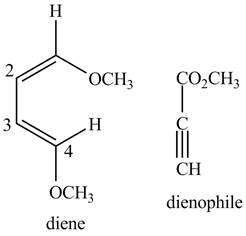
Figure 3
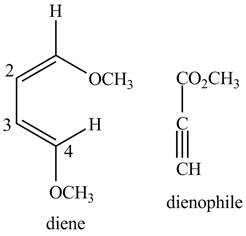
Similarly, second possibility on the basis of above mentioned steps will be as follows.
First designate the double bond carbons as
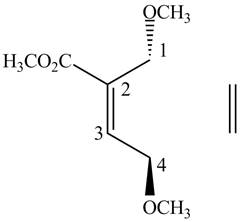
Figure 4
In order to complete the structure of diene, remove the double bonds present between carbons
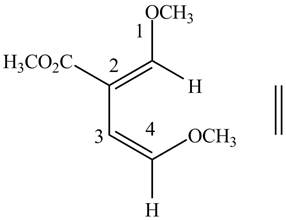
Figure 5
Finally, complete the dienophile structure by adding the double bonds between carbons
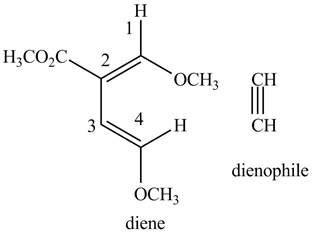
Figure 6
The possible starting materials of the given compound are shown in Figure 3 and Figure 6.
(b)
Interpretation:
The possible structures of the starting materials of the given Diels-Alder product are to be given.
Concept introduction:
First, label the double bond carbons as
In order to complete the diene structure, eliminate the double bonds between
In order to complete the dienophile structure, add the double bonds between its carbons.
Also, dienophile is substituted with electron withdrawing groups.
Answer to Problem 15.20P
The possible structures of starting materials of given compound are shown below.
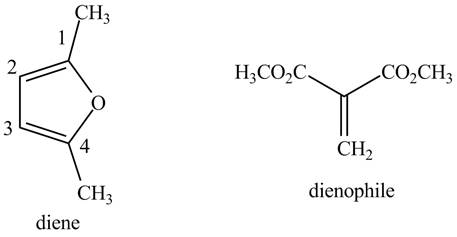
Explanation of Solution
First designate the double bond carbons as
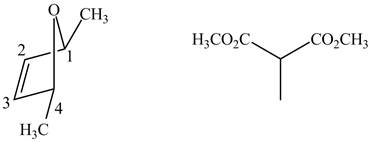
Figure 7
In order to complete the structure of diene, remove the double bonds present between carbons
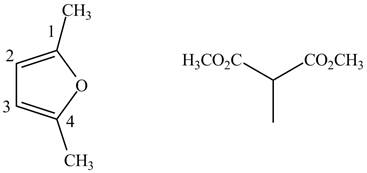
Figure 8
Finally, complete the dienophile structure by adding the double bonds between carbon atoms.
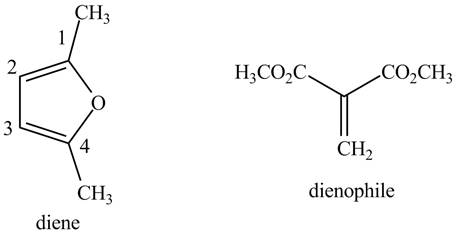
Figure 9
The possible starting materials of given compound are shown in Figure 9.
(c)
Interpretation:
The possible structures of the starting materials of the given Diels-Alder product are to be given.
Concept introduction:
First, label the double bond carbons as
In order to complete the diene structure, eliminate the double bonds between
In order to complete the dienophile structure, add the double bonds between its carbons.
Also, dienophile is substituted with electron withdrawing groups.
Answer to Problem 15.20P
The possible structures of starting materials of given compound are as follows.
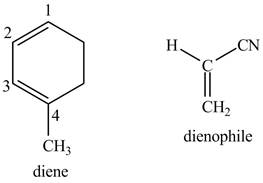
Explanation of Solution
First designate the double bond carbons as
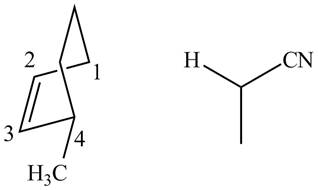
Figure 10
In order to complete the structure of diene, remove the double bonds present between carbons
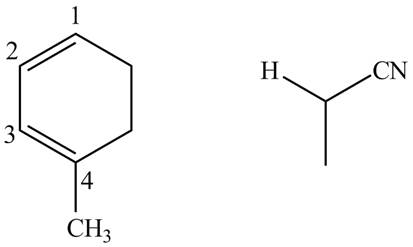
Figure 11
Finally, complete the dienophile structure by adding the double bonds between carbons.
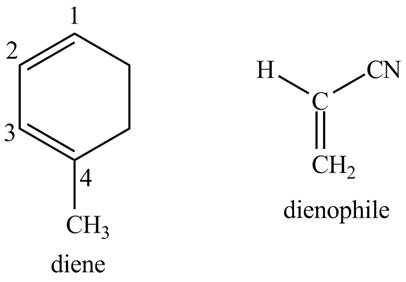
Figure 12
The possible starting materials of given compound are shown in Figure 12.
(d)
Interpretation:
The possible structures of the starting materials of the given Diels-Alder product are to be given.
Concept introduction:
First, label the double bond carbons as
In order to complete the diene structure, eliminate the double bonds between
In order to complete the dienophile structure, add the double bonds between its carbons.
Also, dienophile is substituted with electron withdrawing groups.
Answer to Problem 15.20P
The possible structures of starting materials of given compound are as follows.
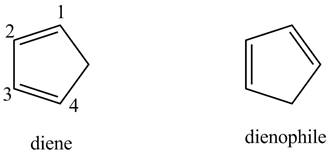
Explanation of Solution
First designate the double bond carbons as
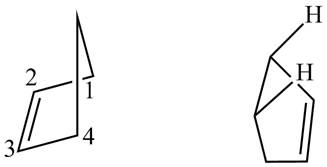
Figure 13
In order to complete the structure of diene, remove the double bonds present between carbons

Figure 14
Finally, complete the dienophile structure by adding the double bonds between carbons.
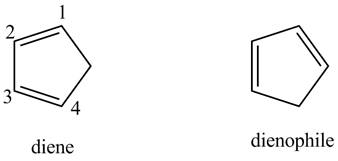
Figure 15
The possible starting materials of given compound are shown in 15.
Want to see more full solutions like this?
Chapter 15 Solutions
ORGANIC CHEMISTRY SAPLING ACCESS + ETEX
- CUE COLUMN NOTES (A. Determine Stereoisomers it has ⑤ Identify any meso B compounds cl Br cl -c-c-c-c-¿- 1 CI C- | 2,4-Dichloro-3-bromopentanearrow_forwardThe acid-base chemistry of both EDTA and EBT are important to ensuring that the reactions proceed as desired, thus the pH is controlled using a buffer. What percent of the EBT indicator will be in the desired HIn2- state at pH = 10.5. pKa1 = 6.2 and pKa2 = 11.6 of EBTarrow_forwardWhat does the phrase 'fit for purpose' mean in relation to analytical chemistry? Please provide examples too.arrow_forward
- For each of the substituted benzene molecules below, determine the inductive and resonance effects the substituent will have on the benzene ring, as well as the overall electron-density of the ring compared to unsubstituted benzene. Molecule Inductive Effects Resonance Effects Overall Electron-Density × NO2 ○ donating O donating O withdrawing O withdrawing O electron-rich electron-deficient no inductive effects O no resonance effects O similar to benzene E [ CI O donating withdrawing O no inductive effects Explanation Check ○ donating withdrawing no resonance effects electron-rich electron-deficient O similar to benzene © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center Accesarrow_forwardUnderstanding how substituents activate Rank each of the following substituted benzene molecules in order of which will react fastest (1) to slowest (4) by electrophilic aromatic substitution. Explanation HN NH2 Check X (Choose one) (Choose one) (Choose one) (Choose one) © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center Aarrow_forwardIdentifying electron-donating and electron-withdrawing effects on benzene For each of the substituted benzene molecules below, determine the inductive and resonance effects the substituent will have on the benzene ring, as well as the overall electron-density of the ring compared to unsubstituted benzene. Inductive Effects Resonance Effects Overall Electron-Density Molecule CF3 O donating O donating O withdrawing O withdrawing O no inductive effects O no resonance effects electron-rich electron-deficient O similar to benzene CH3 O donating O withdrawing O no inductive effects O donating O withdrawing Ono resonance effects O electron-rich O electron-deficient O similar to benzene Explanation Check Х © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Centerarrow_forward
- * Hint: Think back to Chem 1 solubility rules. Follow Up Questions for Part B 12. What impact do the following disturbances to a system at equilibrium have on k, the rate constant for the forward reaction? Explain. (4 pts) a) Changing the concentration of a reactant or product. (2 pts) b) Changing the temperature of an exothermic reaction. (2 pts) ofarrow_forwardDraw TWO general chemical equation to prepare Symmetrical and non-Symmetrical ethers Draw 1 chemical reaction of an etherarrow_forwardPlease help me with the following questions for chemistry.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





