
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 48P
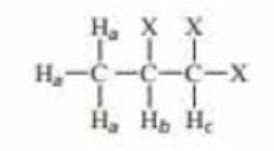
Draw a spitting diagram for the Hb proton and give its multiplicity if
- a. Jba = Jbc.
- b. Jba = 2Jbc.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Predict the major product of the following reaction and then draw a curved arrow mechanism for its formation.
Part: 0/2
Part 1 of 2
H₂SO
heat
: OH
90
Draw the structure of the major product.
Click and drag to start drawing a
structure.
3
Draw a curved arrow mechanism for the reaction, adding steps as necessary. Be sure to include all electrons that are necessary to the mechanism and all
nonzero formal charges.
C
Ö-H
H
+
-S-OH
.0.
Add/Remove step
X
टे
Click and drag to start
drawing a structure.
Draw a curved arrow mechanism for its formation. You may need to re-draw structures to show certain bonds. Ensure that HSO is used as the base to
deprotonate the ẞ carbon when necessary.
C
HO
: OH
HO: OH
=s
=
+
1
Add/Remove step
X
Click and drag to start
drawing a structure.
Chapter 14 Solutions
Organic Chemistry (8th Edition)
Ch. 14.1 - Prob. 1PCh. 14.1 - Prob. 2PCh. 14.4 - How many signals would you expect to see in the 1H...Ch. 14.4 - How many signals would you expect to see in the 1H...Ch. 14.4 - How could you distinguish the 1H NMR spectra of...Ch. 14.4 - Draw an isomer of dichlorocyclopropane that gives...Ch. 14.5 - Prob. 7PCh. 14.5 - Prob. 8PCh. 14.5 - Prob. 9PCh. 14.5 - Where would you expect to find the 1H NMR signal...
Ch. 14.6 - Prob. 11PCh. 14.7 - Prob. 12PCh. 14.7 - Prob. 13PCh. 14.7 - Without referring to Table 14.1, label the proton...Ch. 14.8 - [18]-Annulene shows two signals in its 1H NMR...Ch. 14.9 - How would integration distinguish the 1H NMR...Ch. 14.9 - Which of the following compounds is responsible...Ch. 14.10 - Prob. 19PCh. 14.10 - Explain how the following compounds, each with the...Ch. 14.10 - The 1H NMR spectra of two carboxylic acids with...Ch. 14.11 - Draw a diagram like the one shown in Figure 14.12...Ch. 14.12 - Indicate the number of signals and the...Ch. 14.12 - Explain the relative chemical shifts of the...Ch. 14.12 - How can their 1H NMR spectra distinguish the...Ch. 14.12 - Identify each compound from its molecular formula...Ch. 14.12 - Predict the splitting patterns for the signals...Ch. 14.12 - Describe the 1H NMR spectrum you would expect for...Ch. 14.12 - Propose structures that are consistent with the...Ch. 14.13 - Prob. 30PCh. 14.13 - Identify the compound with molecular formula...Ch. 14.14 - Prob. 32PCh. 14.15 - a. For the following compounds, which pairs of...Ch. 14.15 - How would the 1H NMR spectra for the four...Ch. 14.17 - Explain why the chemical shift of the OH proton of...Ch. 14.17 - Prob. 38PCh. 14.17 - Prob. 39PCh. 14.17 - Prob. 40PCh. 14.20 - Answer the following questions for each compound:...Ch. 14.20 - Prob. 42PCh. 14.20 - How can 1,2-, 1,3-, and 1,4-dinitrobenzene be...Ch. 14.20 - Identify each compound below from its molecular...Ch. 14.22 - Prob. 45PCh. 14.22 - What does cross peak X in Figure 14.34 tell you?Ch. 14 - Prob. 47PCh. 14 - Draw a spitting diagram for the Hb proton and give...Ch. 14 - Label each set of chemically equivalent protons,...Ch. 14 - Determine the ratios of the chemically...Ch. 14 - How can 1H NMR distinguish between the compounds...Ch. 14 - Prob. 52PCh. 14 - Match each of the 1H NMR spectra with one of the...Ch. 14 - The 1H NMR spectra of three isomers with molecular...Ch. 14 - Prob. 55PCh. 14 - Prob. 56PCh. 14 - Compound A, with molecular formula C4H9Cl, shows...Ch. 14 - Would it be better to use 1H NMR or 13C NMR...Ch. 14 - There are four esters with molecular formula...Ch. 14 - Identify the compound with molecular formula C6H14...Ch. 14 - An alkyl halide reacts with an alkoxide ion to...Ch. 14 - The 1H NMR spectra of three isomers with molecular...Ch. 14 - Identity each of the following compounds from its...Ch. 14 - Identity each of the following compounds from its...Ch. 14 - Prob. 65PCh. 14 - How can the signals in the 6.5 to 8.1 ppm region...Ch. 14 - The 1H NMR spectra of two compounds, each with...Ch. 14 - Draw a splitting diagram for the Hb proton if Jbc...Ch. 14 - Sketch the following spectra that would be...Ch. 14 - How can 1H NMR be used to prove that the addition...Ch. 14 - Identity each of the following compounds from its...Ch. 14 - Dr. N. M. Arr was called in to help analyze the 1H...Ch. 14 - Calculate the amount of energy (in calories)...Ch. 14 - The following 1H NMR spectra are four compounds,...Ch. 14 - When compound A (C5H12O) is treated with HBr, it...Ch. 14 - Identity the compound with molecular formula...Ch. 14 - Identity each of the following compounds from its...Ch. 14 - Prob. 78PCh. 14 - Identify each of the following compounds from its...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following could 1,2-ethanediol be directly synthesized from? OH HO О 0 0. O ?arrow_forwardDesign a synthesis of 1,2-diethoxyethane from an alkene. Select the single best answer for each part. Part: 0/3 Part 1 of 3 Which of the following could 1,2-diethoxyethane be directly synthesized from? O HO 0 HO.... OH HO HO × 5 > ?arrow_forwardDraw the skeletal structure of the major organic product of each step of the reaction sequence. Part: 0/2 Part 1 of 2 Part: 1/2 Part 2 of 2 Continue OH NaH Na Na Br + Click and drag to start drawing a structure. X : X G : Garrow_forward
- pleasearrow_forwardplease help me please pleasearrow_forwardUsing reaction free energy to predict equilibrium composition Consider the following equilibrium: N2 (g) + 3H2 (g) = 2NH3 (g) AG⁰ = -34. KJ Now suppose a reaction vessel is filled with 8.06 atm of nitrogen (N2) and 2.58 atm of ammonia (NH3) at 106. °C. Answer the following questions about this system: ? rise Under these conditions, will the pressure of N2 tend to rise or fall? ☐ x10 fall Is it possible to reverse this tendency by adding H₂? In other words, if you said the pressure of N2 will tend to rise, can that be changed to a tendency to fall by adding H₂? Similarly, if you said the pressure of N2 will tend to fall, can that be changed to a tendency to rise by adding H₂? If you said the tendency can be reversed in the second question, calculate the minimum pressure of H₂ needed to reverse it. Round your answer to 2 significant digits. yes no ☐ atm ☑ 5 00. 18 Ararrow_forward
- i need help with the followingarrow_forwardUsing reaction free energy to predict equilibrium composition Consider the following equilibrium: 2NO(g) +Cl₂ (g) = 2NOC1 (g) AGº = -41. kJ Now suppose a reaction vessel is filled with 8.90 atm of chlorine (C12) and 5.71 atm of nitrosyl chloride (NOC1) at 1075. °C. Answer the following questions about this system: rise Under these conditions, will the pressure of NOCI tend to rise or fall? x10 fall Is it possible to reverse this tendency by adding NO? In other words, if you said the pressure of NOCI will tend to rise, can that be changed to a tendency to fall by adding NO? Similarly, if you said the pressure of NOCI will tend to fall, can that be changed to a tendency to rise by adding NO? yes no If you said the tendency can be reversed in the second question, calculate the minimum pressure of NO needed to reverse it. Round your answer to 2 significant digits. atm ☑ 18 Ararrow_forwardIdentifying the major species in weak acid or weak base equilibria The preparations of two aqueous solutions are described in the table below. For each solution, write the chemical formulas of the major species present at equilibrium. You can leave out water itself. Write the chemical formulas of the species that will act as acids in the 'acids' row, the formulas of the species that will act as bases in the 'bases' row, and the formulas of the species that will act as neither acids nor bases in the 'other' row. You will find it useful to keep in mind that HCN is a weak acid. acids: 0.29 mol of NaOH is added to 1.0 L of a 1.2M HCN solution. bases: ☑ other: 0.09 mol of HCl is added to acids: 1.0 L of a solution that is bases: 0.3M in both HCN and KCN. other: 0,0,... ? 00. 18 Ar 日arrow_forward
- Identifying the major species in weak acid or weak base equilibria The preparations of two aqueous solutions are described in the table below. For each solution, write the chemical formulas of the major species present at equilibrium. You can leave out water itself. Write the chemical formulas of the species that will act as acids in the 'acids' row, the formulas of the species that will act as bases in the 'bases' row, and the formulas of the species that will act as neither acids nor bases in the 'other' row. You will find it useful to keep in mind that HF is a weak acid. acids: 0.2 mol of KOH is added to 1.0 L of a 0.5 M HF solution. bases: Х other: ☐ acids: 0.10 mol of HI is added to 1.0 L of a solution that is 1.4M in both HF and NaF. bases: other: ☐ 0,0,... ด ? 18 Ararrow_forwardIdentifying the major species in weak acid or weak base equilibria The preparations of two aqueous solutions are described in the table below. For each solution, write the chemical formulas of the major species present at equilibrium. You can leave out water itself. Write the chemical formulas of the species that will act as acids in the 'acids' row, the formulas of the species that will act as bases in the 'bases' row, and the formulas of the species that will act as neither acids nor bases in the 'other' row. You will find it useful to keep in mind that NH3 is a weak base. acids: ☐ 1.8 mol of HCl is added to 1.0 L of a 1.0M NH3 bases: ☐ solution. other: ☐ 0.18 mol of HNO3 is added to 1.0 L of a solution that is 1.4M in both NH3 and NH₁Br. acids: bases: ☐ other: ☐ 0,0,... ? 000 18 Ar B 1arrow_forwardUsing reaction free energy to predict equilibrium composition Consider the following equilibrium: 2NH3 (g) = N2 (g) +3H₂ —N2 (g) AGº = 34. kJ Now suppose a reaction vessel is filled with 4.19 atm of ammonia (NH3) and 9.94 atm of nitrogen (N2) at 378. °C. Answer the following questions about this system: rise Under these conditions, will the pressure of NH 3 tend to rise or fall? ☐ x10 fall Х Is it possible to reverse this tendency by adding H₂? In other words, if you said the pressure of NH 3 will tend to rise, can that be changed to a tendency to fall by adding H₂? Similarly, if you said the pressure of NH3 will tend to fall, can that be changed to a tendency to rise by adding H₂? If you said the tendency can be reversed in the second question, calculate the minimum pressure of H₂ needed to reverse it. Round your answer to 2 significant digits. yes no atm 00. 18 Ar 무ㅎ ?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY