
Concept explainers
(a)
Interpretation:
The product for the given set of reactions should be identified.
Concept introduction:
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms. The oxidation of alcohol are achieved by using reagents like
(b)
Interpretation:
The product for the given set of reactions should be identified.
Concept introduction:
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms. The oxidation of alcohol are achieved by using reagents like
(c)
Interpretation:
The product for the given set of reactions should be identified.
Concept introduction:
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms. The oxidation of alcohol are achieved by using reagents like
(d)
Interpretation:
The product for the given set of reactions should be identified.
Concept introduction:
Swern oxidation:
Primary and secondary alcohols undergoes oxidation reaction using dimethyl sulfoxide, oxalyl chloride and triethyl
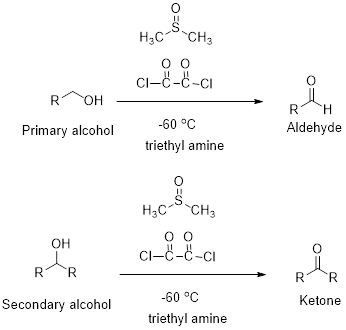
(e)
Interpretation:
The product for the given set of reactions should be identified.
Concept introduction:
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms. The oxidation of alcohol are achieved by using reagents like
(f)
Interpretation:
The product for the given set of reactions should be identified.
Concept introduction:
Oxidation Reaction: The Dess-Martin Periodinane (DMP), a hyper valent iodine compound, offers selective and very mild oxidation of alcohols to aldehydes.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Organic Chemistry
- An electrode process takes place at a metal-solution interface. Indicate the current condition that must be met for Faradaic rectification to occur.arrow_forwardAt a metal-solution interface, an electron is exchanged, and the symmetry factor beta < 0.5 is found in the Butler-Volmer equation. What does this indicate?arrow_forwardTopic: Photochemistry and Photophysics of Supramoleculesarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





