
CONCEPTUAL INTEGRATED SCIENCE (PEARSON+
3rd Edition
ISBN: 2818440059230
Author: Hewitt
Publisher: PEARSON+
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 12, Problem 96TE
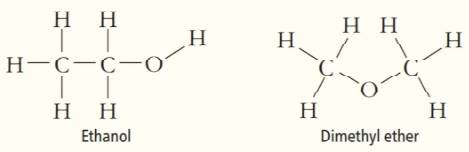
Account for the observation that ethanol, C2H5OH, dissolves readily in water but dimethyl ether, CH3OCH3, which has the same number and kinds of atoms, does not.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For each of the actions depicted, determine the direction (right, left, or zero) of the current induced to flow through the resistor in the circuit containing the secondary coil. The coils are wrapped around a plastic core. Immediately after the switch is closed, as shown in the figure, (Figure 1) in which direction does the current flow through the resistor? If the switch is then opened, as shown in the figure, in which direction does the current flow through the resistor? I have the answers to the question, but would like to understand the logic behind the answers. Please show steps.
When violet light of wavelength 415 nm falls on a single slit, it creates a central diffraction peak that is 8.60
cm wide on a screen that is 2.80 m away.
Part A
How wide is the slit?
ΟΙ ΑΣΦ
?
D= 2.7.10-8
Submit Previous Answers Request Answer
× Incorrect; Try Again; 8 attempts remaining
m
Two complex values are z1=8 + 8i, z2=15 + 7 i. z1∗ and z2∗ are the complex conjugate values.
Any complex value can be expessed in the form of a+bi=reiθ. Find θ for (z1-z∗2)/z1+z2∗. Find r and θ for (z1−z2∗)z1z2∗ Please show all steps
Chapter 12 Solutions
CONCEPTUAL INTEGRATED SCIENCE (PEARSON+
Ch. 12 - How many electrons can occupy the first shell? How...Ch. 12 - Which electrons are represented by an electron-dot...Ch. 12 - Prob. 3RCCCh. 12 - How does an ion differ from an atom?Ch. 12 - To become a negative ion, does an atom lose or...Ch. 12 - Prob. 6RCCCh. 12 - Prob. 7RCCCh. 12 - Prob. 8RCCCh. 12 - Prob. 9RCCCh. 12 - Prob. 10RCC
Ch. 12 - Prob. 11RCCCh. 12 - Prob. 12RCCCh. 12 - Why do nonpolar substances boil at relatively low...Ch. 12 - Which has a greater degree of symmetry-a polar...Ch. 12 - Why dont oil and water mix?Ch. 12 - Prob. 16RCCCh. 12 - What is a hydrogen bond?Ch. 12 - Are induced dipoles permanent?Ch. 12 - What happens to the volume of a sugar solution as...Ch. 12 - Prob. 20RCCCh. 12 - Is concentration typically given with the volume...Ch. 12 - Why does the solubility of a gas solute in a...Ch. 12 - Why do sugar crystals dissolve faster when...Ch. 12 - Is sugar a polar or nonpolar substance?Ch. 12 - Do metals more readily gain or lose electrons?Ch. 12 - What is an alloy?Ch. 12 - What is a native metal?Ch. 12 - Prob. 28TISCh. 12 - Prob. 29TISCh. 12 - How is a solution different from a suspension?Ch. 12 - Prob. 36TCCh. 12 - Prob. 37TCCh. 12 - Rank the following in order of increasing...Ch. 12 - Rank the following in order of decreasing boiling...Ch. 12 - Rank these solutions in order of increasing...Ch. 12 - Rank the following compounds in order of...Ch. 12 - Prob. 42TSCh. 12 - Prob. 43TSCh. 12 - Prob. 44TSCh. 12 - How much sodium chloride, in grams, is needed to...Ch. 12 - If water is added to 1mole of sodium chloride in a...Ch. 12 - Prob. 47TECh. 12 - Prob. 48TECh. 12 - How many more electrons can fit within the valence...Ch. 12 - Prob. 50TECh. 12 - What happens when hydrogens electron gets close to...Ch. 12 - Why does an atom with few valence electrons tend...Ch. 12 - Why it is so easy for a magnesium atom to lose two...Ch. 12 - Why doesnt the neon atom tend to lose or gain any...Ch. 12 - Sulfuric acid, H2SO4, loses two protons to form...Ch. 12 - Prob. 56TECh. 12 - Which should be more difficult to pull apart: a...Ch. 12 - Given that the total number of atoms on our planet...Ch. 12 - Prob. 59TECh. 12 - Two fluorine atoms join together to form a...Ch. 12 - How are metallic bonds similar to ionic bonds? How...Ch. 12 - What drives an atom to form a covalent bond: its...Ch. 12 - Atoms of nonmetallic elements form covalent bonds,...Ch. 12 - Prob. 64TECh. 12 - Prob. 65TECh. 12 - Prob. 66TECh. 12 - In each molecule, which atom carries the greater...Ch. 12 - Which is more polar: a sulfur-bromineS-Br bond or...Ch. 12 - True or False: The greater the nuclear charge of...Ch. 12 - True or False: The more shells in an atom, the...Ch. 12 - Water, H2O, and methane, CH4, have about the same...Ch. 12 - Circle the molecule from each pair that should...Ch. 12 - Three kids sitting equally apart around a table...Ch. 12 - Why is the oxygen atom of a water molecule...Ch. 12 - Look to the molecules listed in Table 12.2. How...Ch. 12 - Which is stronger: the covalent bond that holds...Ch. 12 - The charges with sodium chloride are all...Ch. 12 - Prob. 78TECh. 12 - Prob. 79TECh. 12 - Why is calcium fluoride, CaF2, a high melting...Ch. 12 - Of the two structures shown here, one is a typical...Ch. 12 - Mixtures can be separated into their components by...Ch. 12 - Why cant the elements of a compound be separated...Ch. 12 - Many dry cereals are fortified with iron, which is...Ch. 12 - Classify the following as element, compound, or...Ch. 12 - Which of the following boxes best represents a...Ch. 12 - Which is more dense: air saturated with water...Ch. 12 - How many sugar molecules are there in a 2M sugar...Ch. 12 - Prob. 89TECh. 12 - Which should weigh more: 100mL of fresh water or...Ch. 12 - Prob. 91TECh. 12 - The boiling point of 1, 4-butanediol is 230C....Ch. 12 - Based on atomic size, which would you expect to be...Ch. 12 - If nitrogen, N2, were pumped into your lungs at...Ch. 12 - Prob. 95TECh. 12 - Account for the observation that ethanol, C2H5OH,...Ch. 12 - At 10C, which is more concentrated: a saturated...Ch. 12 - Why is rain or snow called precipitation?Ch. 12 - Hydrogen chloride HCl is a gas at room...Ch. 12 - Some bottled water is now advertised as containing...Ch. 12 - Two plastic bottles of fresh seltzer water are...Ch. 12 - Would you expect to find more dissolved oxygen in...Ch. 12 - What should be done with mining pits after all...Ch. 12 - What are some of the obstacles people face when...Ch. 12 - Oxygen, O2, dissolves quite well within a class of...Ch. 12 - Prob. 1RATCh. 12 - Prob. 2RATCh. 12 - Why are ores so valuable? a They are sources of...Ch. 12 - In terms of the periodic table, is there an abrupt...Ch. 12 - When nitrogen and fluorine combine to form a...Ch. 12 - Prob. 6RATCh. 12 - Someone argues that you shouldnt drink tap water...Ch. 12 - Prob. 8RATCh. 12 - Fish dont live very longer in water that has just...Ch. 12 - Prob. 10RAT
Additional Science Textbook Solutions
Find more solutions based on key concepts
[14.110] The following mechanism has been proposed for the gas-phase reaction of chloroform (CHCI3) and chlorin...
Chemistry: The Central Science (14th Edition)
1.3 Obtain a bottle of multivitamins and read the list of ingredients. What are four chemicals from the list?
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
With what geologic feature are the earthquakes in the mid-Atlantic associated?
Applications and Investigations in Earth Science (9th Edition)
The genotype of F1, individuals in a tetrahybrid cross is AaBbCcDd. Assuming lndependent assortment of these fo...
Campbell Biology (11th Edition)
Which type of cartilage is most plentiful in the adult body?
Anatomy & Physiology (6th Edition)
All of the following terms can appropriately describe humans except: a. primary consumer b. autotroph c. hetero...
Human Biology: Concepts and Current Issues (8th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Calculate the center of mass of the hollow cone shown below. Clearly specify the origin and the coordinate system you are using. Z r Y h Xarrow_forward12. If all three collisions in the figure below are totally inelastic, which will cause more damage? (think about which collision has a larger amount of kinetic energy dissipated/lost to the environment? I m II III A. I B. II C. III m m v brick wall ע ע 0.5v 2v 0.5m D. I and II E. II and III F. I and III G. I, II and III (all of them) 2marrow_forwardCan you solve this 2 question teach me step by step and draw for mearrow_forward
- From this question and answer can you explain how get (0,0,5) and (5,0,,0) and can you teach me how to solve thisarrow_forwardCan you solve this 2 question and teach me using ( engineer method formula)arrow_forward11. If all three collisions in the figure below are totally inelastic, which brings the car of mass (m) on the left to a halt? I m II III m m ע ע ע brick wall 0.5v 2m 2v 0.5m A. I B. II C. III D. I and II E. II and III F. I and III G. I, II and III (all of them)arrow_forward
- How can you tell which vowel is being produced here ( “ee,” “ah,” or “oo”)? Also, how would you be able to tell for the other vowels?arrow_forwardYou want to fabricate a soft microfluidic chip like the one below. How would you go about fabricating this chip knowing that you are targeting a channel with a square cross-sectional profile of 200 μm by 200 μm. What materials and steps would you use and why? Disregard the process to form the inlet and outlet. Square Cross Sectionarrow_forward1. What are the key steps involved in the fabrication of a semiconductor device. 2. You are hired by a chip manufacturing company, and you are asked to prepare a silicon wafer with the pattern below. Describe the process you would use. High Aspect Ratio Trenches Undoped Si Wafer P-doped Si 3. You would like to deposit material within a high aspect ratio trench. What approach would you use and why? 4. A person is setting up a small clean room space to carry out an outreach activity to educate high school students about patterning using photolithography. They obtained a positive photoresist, a used spin coater, a high energy light lamp for exposure and ordered a plastic transparency mask with a pattern on it to reduce cost. Upon trying this set up multiple times they find that the full resist gets developed, and they are unable to transfer the pattern onto the resist. Help them troubleshoot and find out why pattern of transfer has not been successful. 5. You are given a composite…arrow_forward
- Two complex values are z1=8 + 8i, z2=15 + 7 i. z1∗ and z2∗ are the complex conjugate values. Any complex value can be expessed in the form of a+bi=reiθ. Find r and θ for (z1-z∗2)/z1+z2∗. Find r and θ for (z1−z2∗)z1z2∗ Please show all stepsarrow_forwardAn electromagnetic wave is traveling through vacuum in the positive x direction. Its electric field vector is given by E=E0sin(kx−ωt)j^,where j^ is the unit vector in the y direction. If B0 is the amplitude of the magnetic field vector, find the complete expression for the magnetic field vector B→ of the wave. What is the Poynting vector S(x,t), that is, the power per unit area associated with the electromagnetic wave described in the problem introduction? Give your answer in terms of some or all of the variables E0, B0, k, x, ω, t, and μ0. Specify the direction of the Poynting vector using the unit vectors i^, j^, and k^ as appropriate. Please explain all stepsarrow_forwardAnother worker is performing a task with an RWL of only 9 kg and is lifting 18 kg, giving him an LI of 2.0 (high risk). Questions:What is the primary issue according to NIOSH?Name two factors of the RWL that could be improved to reduce risk.If the horizontal distance is reduced from 50 cm to 30 cm, how does the HM change and what effect would it have?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning


Modern Physics
Physics
ISBN:9781111794378
Author:Raymond A. Serway, Clement J. Moses, Curt A. Moyer
Publisher:Cengage Learning

An Introduction to Physical Science
Physics
ISBN:9781305079137
Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:Cengage Learning
The Laws of Thermodynamics, Entropy, and Gibbs Free Energy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=8N1BxHgsoOw;License: Standard YouTube License, CC-BY