
College Physics: A Strategic Approach (4th Edition)
4th Edition
ISBN: 9780134609034
Author: Randall D. Knight (Professor Emeritus), Brian Jones, Stuart Field
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 21CQ
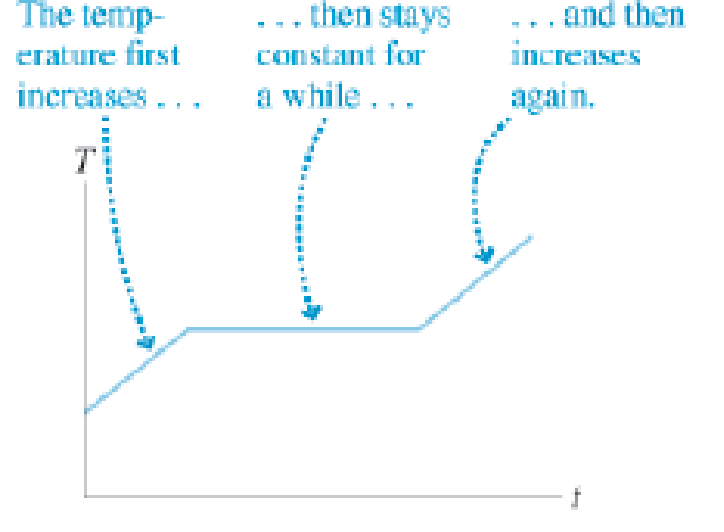
A student is heating chocolate in a pan on the stove. He uses a cooking thermometer to measure the temperature of the chocolate and sees it varies as shown in Figure Q12.21 on the next page. Describe what is happening to the chocolate in each of the three portions of the graph.
Figure Q12.21
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
An experiment was performed to measure the thermal conductivity of food. A large 5 mm
thick slice of the food was cut and the temperature measured separately on each side of the
piece. When the temperature difference through the food was kept constant at 35 ° C, the
heat flow per area was measured and turned out to be 4700 W/ m2. Calculate the thermal
conductivity of the food and explain the conclusions you use in the calculations.
A solid metal cube has a length of 1 meter and across sectional area of 1 m2. What does it mean that the thermal conductivity of the metal is 170 W/(m °C)?
a. 170 W of heat would flow through the long direction if the block were 170 m long with a temperature difference of 1°C between its two ends.
b. 1 W of heat flows per 170 m2 cross sectional area when the temperature difference is 1°C.
c. It takes 170°C of temperature difference to cause1 W of heat to flow through the cube.
d. 170 W of heat flows through the cube when the temperature difference between the two sides is 1°C.
e. t takes 170 s for 1 W of heat to flow through the cube when the temperature difference is 1°C.
The specific heat at constant volume of a particular gas is 0.182 kcal/kg.k at room temperature and it's molecular mass is 34. a) what is its specific heat at constant pressure? b) what do you think is the molecular structure of this gas?
Chapter 12 Solutions
College Physics: A Strategic Approach (4th Edition)
Ch. 12 - Which has more mass, a mole of Ne gas or a mole of...Ch. 12 - If you launch a projectile upward with a high...Ch. 12 - Prob. 3CQCh. 12 - If you double the typical speed of the molecules...Ch. 12 - Two gases have the same number of molecules per...Ch. 12 - If the temperature T of an ideal gas doubles, by...Ch. 12 - A bottle of helium gas and a bottle of argon gas...Ch. 12 - A gas cylinder contains 1.0 mol of helium at a...Ch. 12 - Prob. 9CQCh. 12 - Prob. 10CQ
Ch. 12 - You need to precisely measure the dimensions of a...Ch. 12 - A common trick for opening a stubborn lid on a jar...Ch. 12 - Prob. 13CQCh. 12 - Materials A and B have equal densities, but A has...Ch. 12 - Prob. 15CQCh. 12 - You need to raise the temperature of a gas by 10C....Ch. 12 - Prob. 18CQCh. 12 - Prob. 19CQCh. 12 - A sample of ideal gas is in a cylinder with a...Ch. 12 - A student is heating chocolate in a pan on the...Ch. 12 - If you bake a cake at high elevation, where...Ch. 12 - Prob. 23CQCh. 12 - Prob. 24CQCh. 12 - Prob. 25CQCh. 12 - Prob. 26CQCh. 12 - Prob. 27CQCh. 12 - Prob. 29CQCh. 12 - Prob. 30MCQCh. 12 - Prob. 31MCQCh. 12 - A gas is compressed by an isothermal process that...Ch. 12 - Prob. 33MCQCh. 12 - Prob. 34MCQCh. 12 - Prob. 35MCQCh. 12 - Prob. 36MCQCh. 12 - Prob. 37MCQCh. 12 - Prob. 38MCQCh. 12 - Prob. 1PCh. 12 - How many grams of water (H2O) have the same number...Ch. 12 - Prob. 3PCh. 12 - How many cubic millimeters (mm3) are in 1 L?Ch. 12 - Prob. 5PCh. 12 - Prob. 6PCh. 12 - Prob. 7PCh. 12 - Prob. 8PCh. 12 - An ideal gas is at 20C. The gas is cooled,...Ch. 12 - An ideal gas at 0C consists of 1.0 1023 atoms. 10...Ch. 12 - An ideal gas at 20C consists of 2.2 1022 atoms....Ch. 12 - Prob. 12PCh. 12 - Prob. 13PCh. 12 - When you stifle a sneeze, you can damage delicate...Ch. 12 - Prob. 15PCh. 12 - Mars has an atmosphere composed almost entirely of...Ch. 12 - Prob. 18PCh. 12 - The lowest pressure ever obtained in a laboratory...Ch. 12 - Prob. 20PCh. 12 - Helium has the lowest condensation point of any...Ch. 12 - Prob. 22PCh. 12 - Prob. 23PCh. 12 - Prob. 24PCh. 12 - Prob. 25PCh. 12 - Prob. 26PCh. 12 - A cylinder contains 3.0 L of oxygen at 300 K and...Ch. 12 - Prob. 28PCh. 12 - 0.10 mol of argon gas is admitted to an evacuated...Ch. 12 - Prob. 30PCh. 12 - 0.10 mol of argon gas is admitted to an evacuated...Ch. 12 - 0.10 mol of argon gas is admitted to an evacuated...Ch. 12 - Prob. 33PCh. 12 - Prob. 34PCh. 12 - Prob. 35PCh. 12 - Prob. 36PCh. 12 - A 1.0 cm3 air bubble is released from the sandy...Ch. 12 - A weather balloon rises through the atmosphere,...Ch. 12 - Prob. 39PCh. 12 - Prob. 40PCh. 12 - Prob. 41PCh. 12 - Prob. 42PCh. 12 - The length of a steel beam increases by 0.73 mm...Ch. 12 - Prob. 44PCh. 12 - The length of a steel beam increases by 0.73 mm...Ch. 12 - At 20C, the hole in an aluminum ring is 2.500 cm...Ch. 12 - The temperature of an aluminum disk is increased...Ch. 12 - Prob. 48PCh. 12 - Prob. 49PCh. 12 - Prob. 50PCh. 12 - a. 100 J of heat energy are transferred to 20 g of...Ch. 12 - Prob. 52PCh. 12 - Prob. 53PCh. 12 - Prob. 54PCh. 12 - Alligators and other reptiles dont use enough...Ch. 12 - Prob. 56PCh. 12 - When air is inhaled, it quickly becomes saturated...Ch. 12 - Prob. 58PCh. 12 - Prob. 59PCh. 12 - What minimum heat is needed to bring 100 g of...Ch. 12 - Brewed coffee is often too hot to drink right...Ch. 12 - Prob. 62PCh. 12 - 30 g of copper pellets are removed from a 300C...Ch. 12 - Prob. 64PCh. 12 - A copper block is removed from a 300C oven and...Ch. 12 - Prob. 66PCh. 12 - If a person has a dangerously high fever,...Ch. 12 - A container holds 1.0 g of argon at a pressure of...Ch. 12 - A container holds 1.0 g of oxygen at a pressure of...Ch. 12 - What is the temperature change of 1.0 mol of a...Ch. 12 - Heating 2.5 mol of neon in a rigid container...Ch. 12 - Prob. 72PCh. 12 - A 1.8-cm-thick wood floor covers a 4.0 m 5.5 m...Ch. 12 - A stainless-steel-bottomed kettle, its bottom 24...Ch. 12 - Seals may cool themselves by using thermal...Ch. 12 - Electronics and inhabitants of the International...Ch. 12 - The glowing filament in a lamp is radiating energy...Ch. 12 - Prob. 78PCh. 12 - If you lie on the ground at night with no cover,...Ch. 12 - Prob. 80PCh. 12 - Prob. 81PCh. 12 - Prob. 82PCh. 12 - Prob. 83GPCh. 12 - Prob. 84GPCh. 12 - Prob. 85GPCh. 12 - Prob. 86GPCh. 12 - Prob. 87GPCh. 12 - A 5.0-m-diameter garden pond holds 5.9 103 kg of...Ch. 12 - James Joule (after whom the unit of energy is...Ch. 12 - Prob. 90GPCh. 12 - Prob. 91GPCh. 12 - Prob. 92GPCh. 12 - A 68 kg woman cycles at a constant 15 km/h. All of...Ch. 12 - Prob. 94GPCh. 12 - Prob. 95GPCh. 12 - Prob. 97GPCh. 12 - Your 300 mL cup of coffee is too hot to drink when...Ch. 12 - A gas is compressed from 600 cm3 to 200 cm3 at a...Ch. 12 - An expandable cube, initially 20 cm on each side,...Ch. 12 - Prob. 101GPCh. 12 - Prob. 102GPCh. 12 - Prob. 103GPCh. 12 - Homes are often insulated with fiberglass...Ch. 12 - The surface area of an adult human is about 1.8...Ch. 12 - Prob. 106MSPPCh. 12 - Prob. 107MSPPCh. 12 - Prob. 108MSPPCh. 12 - Prob. 109MSPPCh. 12 - Prob. 110MSPPCh. 12 - Prob. 111MSPPCh. 12 - Prob. 112MSPP
Additional Science Textbook Solutions
Find more solutions based on key concepts
A block is launched with speed v0 up a slope making an angle with the horizontal; the coefficient of kinetic f...
Essential University Physics: Volume 1 (3rd Edition)
The object distance that is required to give an image with a a magnification of +2.0 .
Physics (5th Edition)
The pV-diagram of the Carnot cycle.
Sears And Zemansky's University Physics With Modern Physics
Sedna Orbit. The object Sedna orbits our Sun at an average distance (semimajor axis) of 509 AU. What is its orb...
Life in the Universe (4th Edition)
9. The forces in FIGURE EX6.9 act on a 2.0 kg object. What are the values of ax and ay, the x- and y-componen...
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Another property of light is the energy. Which of the following has the greatest energy (circle one)?
Lecture- Tutorials for Introductory Astronomy
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- The Sun radiates like a perfect black body with an emissivity of exactly 1. (a) Calculate the surface temperature of the Sun, given that it is a sphere with a 7.00108m radius that radiates 3.801026W into 3-K space. (b) How much power does the Sun radiate per square meter of its surface? (c) How much power in watts per square meter is that value at the distance of Earth, 1.501011m away? (This number is called the solar constant.)arrow_forwardA glass windowpane in a home is 0.620 cm thick and has dimensions of 1.00 in 2.00 in. On a certain day, the temperature of the interior surface of the glass is 25.0C and the exterior surface temperature is 0C. (a) What is the rate at which energy is transferred by heat through the glass? (b) How much energy is transferred through the window in one day, assuming the temperatures on the surfaces remain constant?arrow_forward(a) A firewalker runs across a bed of hot coals without sustaining burns. Calculate the heat transferred by conduction into the sole of one foot of a firewalker given that the bottom of the foot is a 3.00-mm-thick callus with a conductivity at the low end of the range for wood and its density is 300kg/m3. The area of contact is 25.0cm2, the temperature of the coals is 700C, and the time in contact is 1.00 s. (b) What temperature increase is produced in the 25.0cm3 of tissue affected? (c) What effect do you think this will have on the tissue, keeping in mind that a callus is made of dead cells?arrow_forward
- (a) What is the average kinetic energy in joules of hydrogen atoms on the 5500C surface at the Sun? (b) What is the average kinetic energy of helium atoms in a region of the solar corona where me temperature is 6.00105K ?arrow_forwardUnreasonable Results (a) An automobile mechanic claims that an aluminum rod fits loosely into its hole on an aluminum engine block because the engine is hot and the rod is cold. If 1he hole is 10.0% bigger in diameter than the 22.0C rod, at what temperature will the rod be the same size as the hole? (b) What is unreasonable about this temperature? (2) Which premise is responsible?arrow_forwardA grandfather clock is controlled by a swinging brass pendulum that is 1.3 m long at a temperature of 20.0C. (a) What is the length of the pendulum rod when the temperature drops to 0.0C? (b) If a pendulums period is given by T=2L/g, where L is its length, does the change in length of the rod cause the clock to run fast or slow?arrow_forward
- A steel plate has a circular hole drilled in its center. If the diameter of the hole varies according to thermal linear expansion, show that the area of the original circle A0 changes with an increase in temperature T, following the approximate relation A CA0 T, where C = 2. Hint: (T)2T.arrow_forwardFigure P20.45 shows a phase diagram of carbon dioxide in terms of pressure and temperature, a. Use the phase diagram to explain why dry ice (solid carbon dioxide) sublimates into vapor at atmospheric pressure rather than melting into a liquid. At what temperature does the dry ice sublimate when at atmospheric pressure? b. Estimate what pressure would be needed to liquefy carbon dioxide at room temperature.arrow_forwardIt is the morning of a day that will become hot. You just purchased drinks for a picnic and are loading them, with ice, into a chest in the back of your car. (a) You wrap a wool blanket around the chest. Does doing so help to keep the beverages cool, or should you expect the wool blanket to warm them up? Explain your answer. (b) Your younger sister suggests you wrap her up in another wool blanket to keep her cool on the hot day like the ice chest. Explain your response to her.arrow_forward
- In 1986, a gargantuan iceberg broke away from the Ross Ice Shelf in Antarctica. It was approximately a rectangle 160 km long, 40.0 km wide, and 250 m thick. (a) What is the mass of this iceberg, given that the density of ice is 917kg/m3 ? (b) How much heat transfer (in joules) is needed to melt it? (c) How many years would it take sunlight alone to melt ice this thick, if the ice absorbs an average of 100W/m2, 12.00 h per day?arrow_forwardThe distance between telephone poles is 30.50 m in a neighborhood where the temperature ranges from 35C to 40C. If you hang a copper cable between two adjacent poles on a day when the temperature is 22.30C, what is the minimum length of the copper cable you must use for the cable to remain connected to the poles all year? Assume the cable is straight, and ignore the effect of gravity on the cable. Consider to have four significant figures and report your answer to four significant figures.arrow_forward(a) What is the rate of heat conduction through the 3.00-cm-thick fur of a large animal having a 1.40m2 surface area? Assume that the animal’s skin temperature is 32.0C, that tile air temperature is 5.00C, and that fur has the same thermal conductivity as air. (b) What food intake will the animal need in one day to replace this heat transfer?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning
Heat Transfer: Crash Course Engineering #14; Author: CrashCourse;https://www.youtube.com/watch?v=YK7G6l_K6sA;License: Standard YouTube License, CC-BY