
Why do ethanol and dimethyl ether have such different properties even though they have same chemical formula?
To determine: Why do ethanol and dimethyl ether have such different properties even when they have same chemical formula?
Answer to Problem 1E
Solution:
The difference is caused because of different functional group present in both the compounds. Ethanol contains alcohol (-OH) functional group while dimethyl ether contains ether (-O-) group.
Explanation of Solution
The physical and chemical properties of any compound is closely related with its structure and bonding pattern.
So, to understand the difference between properties of the given compounds their structural formula are very important to be discussed.
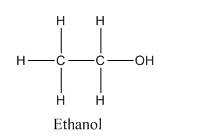
Chemical formula of Ethanol : C2H6O or , C2H5OH
Structural formula of Ethanol :
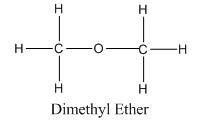
Chemical formula of Dimethyl ether : C2H6O or , CH3-O-CH3
Structural formula of Ethanol :
The difference can be easily seen, that in one hand, where ethanol is a polar molecule with terminal −OH group having electronegativity difference and the ethanol molecules are attached together by hydrogen bonding as the H-atom is directly attached to electronegative O-atom.
While in other hand, dimethyl ether has two methyl groups attached to one other by (-O-) group on either sides which generates a regular dipole-dipole interactions.
The two types of bonding pattern bring changes in the properties of the two compounds.
Also, there are many more functional aspects of both the functional groups which change the properties of compounds instead of having same chemical formula.
The difference in functional groups of ethanol and dimethyl ether brings the difference in their properties.
Want to see more full solutions like this?
Chapter 11 Solutions
Chemistry: Structure and Properties (2nd Edition)
- Reason whether it is possible to determine changes in the Galvani potential difference at the metal-solution interface.arrow_forwardObtain the standard potential at 25°C of the Cu* I Cu | Pt electrode from the standard potentials E° Cu²+/Cu = 0.341 V and E Cu²+ /Cu+ = 0.153 V.arrow_forwardIn electrochemistry, briefly describe the Galvani potential, the Volta potential, and the surface potential. Differentiate between them.arrow_forward
- What substances can neutralize, complex or adsorb and absorb both HF and CF carbonyl fluoride and hydrogen fluoride and intermediate formation of thermal decomposition of fluorinated inorganic compounds either due to hydrolysis and hygroscopic reactions. What is the known chemistry of these reactions and mechanisms.arrow_forwardBriefly differentiate between chemical potential and electrochemical potential.arrow_forwardAccording to open access forums ionic antimony Sb (111) can be reduced to elemental Sb (0) in solution and in macromolecules like condensation polymers polyethylene terephthalate (PET) causing greying of the polymer matrix. It has been connected to thermal degradation of the polymer during processing to the formation of thermally unstable EG ethyleen glycol that forms at various temperatures formic acid, formaldehyde, acetaldehyde and much more depending on temperature. I need to know what organics are more powerful reducing agents and at what concentration (relative) to each organic will initiate this reduction. Furthermore, is the pH dependant ? Are other trace elements in the plastic also a cause of concern e.g. aluminum from aluminum chloride (lewis acid). Therefore, the ultimate solution should include a means to inhibit reduction of ionic antimony and will the same solution comply with cobalt impurities from ionic cobalt? Some PET have combinations of catalyst and their residues…arrow_forward
- From a pH standpoint is the reduction of ionic Antimony Sb (111) to elemental Sb (0) occur more readily by acidic species acting as reducing agents or basic substances? I want to inhibit this reduction of ionic to elemental. Suggestions and directions!arrow_forwardObtain the standard potential at 25°C of the Cu* I Cu | Pt electrode from the standard potentials E° Cu²+/Cu = 0.341 V and E Cu²+ /Cu+ = 0.153 V.arrow_forwardState two variables on which the transport number in electrochemistry depends.arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning



