
(a)
Interpretation:
Given compound has to be named using
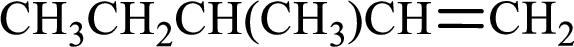
Concept Introduction:
A common nomenclature of naming organic compounds has been developed by IUPAC. By usage of this nomenclature or rules, memorizing of names of organic compounds is not necessary.
IUPAC rules for naming
There are about five rules that has to be followed for naming an alkene and an
- The longest continuous carbon chain in the compound that contains double bond or triple has to be identified. This is known as parent compound.
- Suffix “–ane” (in name of
alkane ) is replaced with “-ene” for alkene or “-yne” for alkyne. - Numbering has to be done so that the lowest number is given to the double or triple bond.
- Naming and numbering has to be given for each atom or group that is attached to the parent chain. Numbering has to be done in a way that substituents get the least numbering.
- If the alkenes have more than one double bond they are called as alkadienes (two double bonds) or alkatrienes (three double bonds). Appropriate suffix has to be used depending on the number of multiple bonds present in the compound.
(a)
Explanation of Solution
Given compound is,
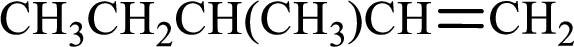
Longest carbon chain with double bond is found to contain five carbon atoms. Therefore, the parent alkane is pentane. As a double bond is present, the alkene name is pentene.
Numbering has to be given in a way that the double bond gets the least numbering. In this case, double bond is present between carbon-1 and carbon-2. Therefore, the parent alkene is 1-pentene.
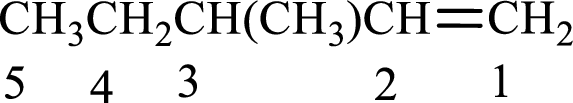
The substituent present in the given structure is a methyl group on carbon-3. Substituents name has to be arranged in the alphabetical order. Number has to be added before the substituent names which indicate the carbon atom in which it is present. This gives the name of alkene as 3-methyl-1-pentene.
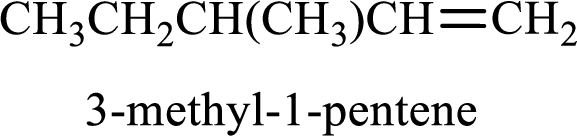
Longest carbon chain containing double bond is pentane. Position of double bond is 1-pentene. Substituents present in the chain are 3-methyl. IUPAC name of the alkene given is 3-methyl-1-pentene.
(b)
Interpretation:
Given compound has to be named using IUPAC nomenclature.
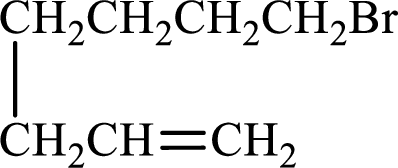
Concept Introduction:
Refer part (a).
(b)
Explanation of Solution
Given compound is,
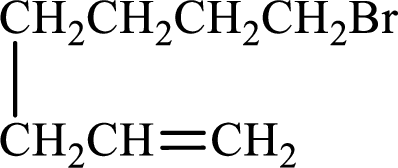
Longest carbon chain with double bond is found to contain seven carbon atoms. Therefore, the parent alkane is heptane. As a double bond is present, the alkene name is heptene.
Numbering has to be given in a way that the double bond gets the least numbering. In this case, double bond is present between carbon-1 and carbon-2. Therefore, the parent alkene is 1-heptene.
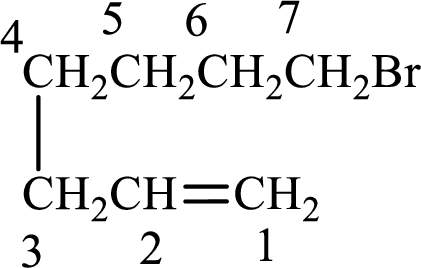
The substituent present in the given structure is a bromine atom on carbon-7. Number has to be added before the substituent names which indicate the carbon atom in which it is present. This gives the name of alkene as 7-methyl-1-heptene.
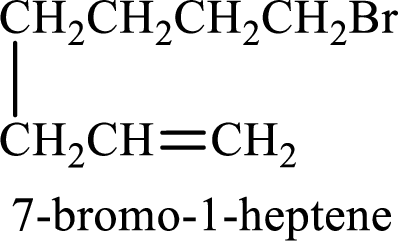
Longest carbon chain containing double bond is heptane. Position of double bond is 1-heptene. Substituents present in the chain are 7-bromo. IUPAC name of the alkene given is 7-bromo-1-heptene.
(c)
Interpretation:
Given compound has to be named using IUPAC nomenclature.
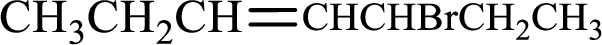
Concept Introduction:
Refer part (a).
(c)
Explanation of Solution
Given compound is,
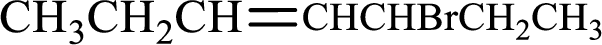
Longest carbon chain with double bond is found to contain seven carbon atoms. Therefore, the parent alkane is heptane. As a double bond is present, the alkene name is heptene.
Numbering has to be given in a way that the double bond gets the least numbering. In this case, double bond is present between carbon-3 and carbon-4. Therefore, the parent alkene is 3-heptene.
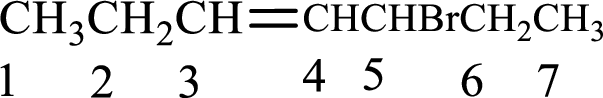
The substituent present in the given structure is a bromine atom on carbon-5. Number has to be added before the substituent names which indicate the carbon atom in which it is present. This gives the name of alkene as 5-bromo-3-heptene.
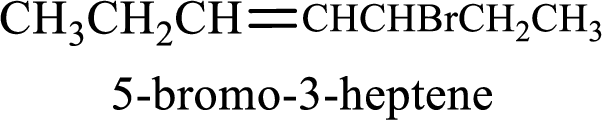
Longest carbon chain containing double bond is heptane. Position of double bond is 3-heptene. Substituents present in the chain are 5-bromo. IUPAC name of the alkene given is 5-bromo-3-heptene.
(d)
Interpretation:
Given compound has to be named using IUPAC nomenclature.
Concept Introduction:
A common nomenclature of naming organic compounds has been developed by IUPAC. By usage of this nomenclature or rules, memorizing of names of organic compounds is not necessary.
IUPAC rules for naming cycloalkenes:
- The number of carbon atoms present in the ring is counted and the name of the alkane that has the same number of carbon atoms is given by adding prefix “cyclo-” to the alkane name. Suffix “-ane” is changed as “-ene”.
- The double bond that is present in the ring is given always the number 1.
- If the ring is substituted, then the names of the group or atoms have to be placed before the name of cycloalkene.
- If the ring contains more than one substituent, then the numbers has to be used in a way that it gives the lowest position for the substituents following position 1 for the double bond.
(d)
Explanation of Solution
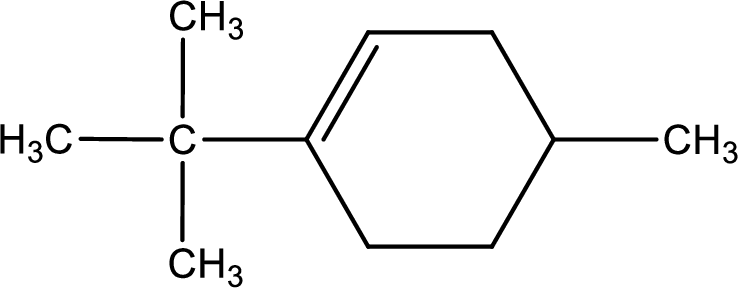
Given compound is,
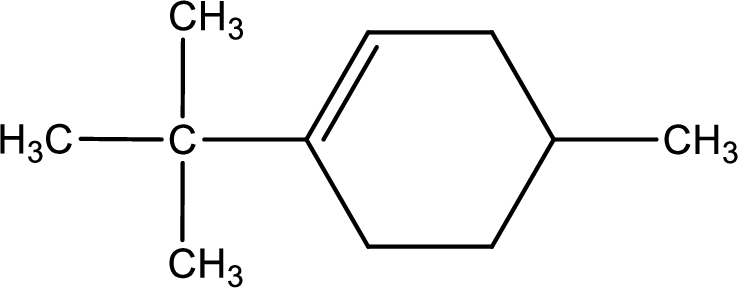
Given compound is found to contain six carbon atoms in a cyclic ring structure. Therefore, the parent cycloalkane is cyclohexane. As there is a double bond present in the ring, the parent compound name is cyclohexene.
Numbering has to be given in a way that the double bond gets the least numbering. In this case, double bond is present between carbon-1 and carbon-2.
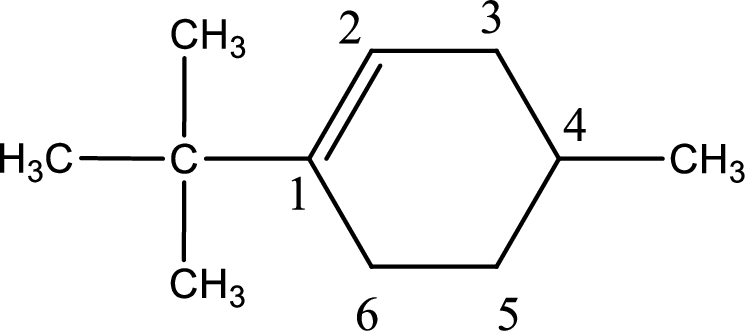
The substituents present in the given structure are a tert-butyl group on carbon-1 and a methyl group on carbon-4. Number has to be added before the substituent names which indicate the carbon atom in which it is present. This gives the name of compound as 1-tert-butyl-4-methylcyclohexene.
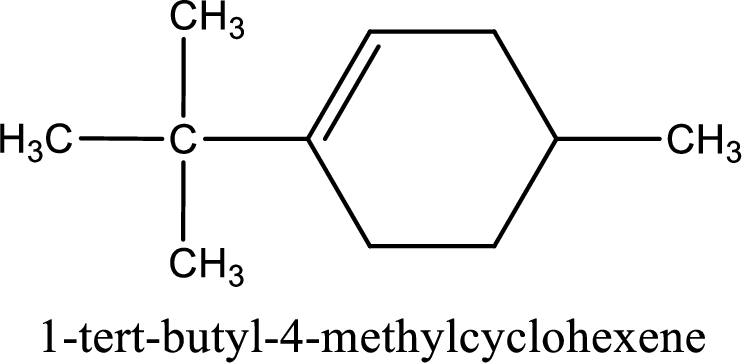
Parent chain is found to be cyclohexene. Position of double bond is carbon-1. Substituent present in the chain is 1-tert-butyl-4-methyl. IUPAC name of the compound given is 1-tert-butyl-4-methylcyclohexene.
Want to see more full solutions like this?
Chapter 11 Solutions
Connect 1-Semester Access Card for General, Organic, and Biochemistry
- x + LEKS: Using a phase diagram a X n/alekscgi/x/lsl.exe/10_u-IgNsikr7j8P3jH-IQs_pBan HhvlTCeeBZbufu BYTI0Hz7m7D3ZcHYUt80XL-5alyVpw ○ States of Matter Using a phase diagram to find a phase transition temperature or pressure Use the phase diagram of Substance X below to find the melting point of X when the pressure above the solid is 1.1 atm. pressure (atm) 16 08- solid liquid- 0 200 400 gas 600 temperature (K) Note: your answer must be within 25 °C of the exact answer to be graded correct. × 5arrow_forwardS: Using a phase diagram leksogi/x/sl.exe/1ou-IgNs kr 7j8P3jH-IQs_pBan HhvTCeeBZbufuBYTI0Hz7m7D3ZdHYU+80XL-5alyVp O States of Matter Using a phase diagram to find a phase transition temperature or pressure se the phase diagram of Substance X below to find the boiling point of X when the pressure on the liquid is 1.6 atm. pressure (atm) 32- 16- solid liquid 0. gas 100 200 temperature (K) 300 Note: your answer must be within 12.5 °C of the exact answer to be graded correct. 10 Explanation Check § Q Search J 2025 McGraw Hill LLC. All Rights Researrow_forward151.2 254.8 85.9 199.6 241.4 87.6 242.5 186.4 155.8 257.1 242.9 253.3 256.0 216.6 108.7 239.0 149.7 236.4 152.1 222.7 148.7 278.2 268.7 234.4 262.7 283.2 143.6 QUESTION: Using this group of data on salt reduced tomato sauce concentration readings answer the following questions: 1. 95% Cl Confidence Interval (mmol/L) 2. [Na+] (mg/100 mL) 3. 95% Na+ Confidence Interval (mg/100 mL)arrow_forward
- Results Search Results Best Free Coursehero Unloc xb Success Confirmation of Q x O Google Pas alekscgi/x/lsl.exe/1o_u-IgNslkr 7j8P3jH-IQs_pBanHhvlTCeeBZbufu BYTI0Hz7m7D3ZcHYUt80XL-5alyVpwDXM TEZayFYCavJ17dZtpxbFD0Qggd1J O States of Matter Using a phase diagram to find a phase transition temperature or pressure Gabr 3/5 he pressure above a pure sample of solid Substance X at 101. °C is lowered. At what pressure will the sample sublime? Use the phase diagram of X below to nd your answer. pressure (atm) 24- 12 solid liquid gas 200 400 temperature (K) 600 ote: your answer must be within 0.15 atm of the exact answer to be graded correct. atm Thanation Check © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center I Q Search L³ ملةarrow_forward301.7 348.9 193.7 308.6 339.5 160.6 337.7 464.7 223.5 370.5 326.6 327.5 336.1 317.9 203.8 329.8 221.9 331.7 211.7 309.6 223.4 353.7 334.6 305.6 340.0 304.3 244.7 QUESTION: Using this group of data on regular tomato sauce concentration readings answer the following questions: 1. 95% Cl Confidence Interval (mmol/L) 2. [Na+] (mg/100 mL) 3. 95% Na+ Confidence Interval (mg/100 mL)arrow_forwardSearch Results Search Results Best Free Coursehero Unlo x b Success Confirmation of Q aleks.com/alekscgi/x/sl.exe/10_u-lgNslkr7j8P3jH-IQs_pBan HhvlTCeeBZbufu BYTIOHz7m7D3ZcHYUt80XL-5alyVpwDXM TEZayFYCav States of Matter Using a phase diagram to find a phase transition temperature or pressure Use the phase diagram of Substance X below to find the temperature at which X turns to a gas, if the pressure above the solid is 3.7 atm. pressure (atm) 0. 32- 16 solid liquid gas 200 temperature (K) Note: your answer must be within 20 °C of the exact answer to be graded correct. Дос Xarrow_forward
- Consider the reaction below to answer the following questions: Acetoacetic ester can be prepared by the Claisen self-condensation reaction of ethyl acetate. 1. NaOEt, EtOH H&C OCH CH3 2 H30 H3C CH2 OCH2CH3 A. Write the complete stepwise mechanism for this reaction. Show all electron flow with arrows and draw all intermediate structures. B. Ethyl acetate can be prepared from ethanol as the only organic starting material. Show all reagents and structures for all intermediates in this preparation. C. Give the structures of the ester precursors for the following Claisen condensation product and formulate the reaction. OEtarrow_forwardUse the phase diagram of Substance X below to find the temperature at which X turns to a gas, if the pressure above the solid is 3.7 atm. pressure (atm) 32 16 solid liquid gas 0 0 200 temperature (K) Note: your answer must be within 20 °C of the exact answer to be graded correct. Шос ☑ كarrow_forwardStarting from bromoethane, how could you prepare the following compounds: a. Ethanol. b. Acetaldehyde f. Acetone. e. 2-Propanol i. Acetoacetic ester. d. 2-Bromoacetic acid. c. Acetic acid g. Acetamide. j. Ethylmalonate k. Gama ketoacid. h. Ethyl magnesium bromide.arrow_forward
- - The pressure above a pure sample of solid Substance X at 60. °C is raised. At what pressure will the sample melt? Use the phase diagram of X below to find your answer. pressure (atm) 02 0.4 solid Hliquid gas 0 0 200 400 600 temperature (K) Note: your answer must be within 0.025 atm of the exact answer to be graded correct. ☐ atmarrow_forward15. What is the order of decreasing reactivity towards nucleophilic acyl substitution for the carboxylic acid derivatives? (most reactive first) 0 O H3C COC CH3 H₂C C N(CH3)2 H3C C OCH3 A. a. I, 11, 111, b. I, III, IV, II C. II, IV, III, I ° (CH3)2CH C OCH3 IV d. II, I, III, IV B. R COCR 0 0 0 13= RC NH2 RC OR RC CI === IV a. I, III, II, IV b. II, III, I, IV C. III, II, I, IV d. IV, I, III, IIarrow_forwardDraw the formula of the product obtained by reacting D-Tallose with bromine water.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





