
Bundle: Chemistry, Loose-Leaf Version, 10th + OWLv2, 4 terms (24 months) Printed Access Card
10th Edition
ISBN: 9781337537933
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 71E
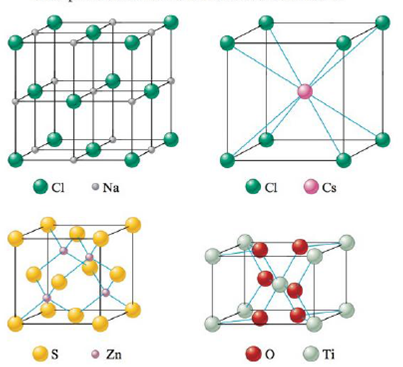
The structures of some common crystalline substances are shown below. Show that the net composition of each unit cell corresponds to the correct formula of each substance.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Select the product for the following reaction.
HO
HO
PCC
OH
○
OH
O HO
○ HO
HO
HO
5:45
Х
Select the final product for the following reaction sequence.
O
O
1. Mg. ether
2.D.O
Based on the chart
Two similarities between the molecule with alpha glycosidic linkages.
Two similarities between the molecules with beta glycosidtic linkages.
Two differences between the alpha and beta glycosidic linkages.
Chapter 10 Solutions
Bundle: Chemistry, Loose-Leaf Version, 10th + OWLv2, 4 terms (24 months) Printed Access Card
Ch. 10 - What are intermolecular forces? How do they differ...Ch. 10 - Define the following terms and describe how each...Ch. 10 - Compare and contrast solids, liquids, and gases.Ch. 10 - Prob. 4RQCh. 10 - What is a lattice? What is a unit cell? Describe a...Ch. 10 - What is closest packing? What is the difference...Ch. 10 - Describe, in general, the structures of ionic...Ch. 10 - Prob. 9RQCh. 10 - Prob. 10RQCh. 10 - Compare and contrast the phase diagrams of water...
Ch. 10 - It is possible to balance a paper clip on the...Ch. 10 - Consider a sealed container half-filled with...Ch. 10 - Explain the following: You add 100 mL water to a...Ch. 10 - Prob. 4ALQCh. 10 - Prob. 5ALQCh. 10 - Why do liquids have a vapor pressure? Do all...Ch. 10 - Prob. 7ALQCh. 10 - What is the vapor pressure of water at 100C? How...Ch. 10 - Prob. 9ALQCh. 10 - Prob. 10ALQCh. 10 - Prob. 11ALQCh. 10 - Why is N2 a gas at room temperature? Explain why...Ch. 10 - Prob. 13ALQCh. 10 - In the diagram below, which lines represent the...Ch. 10 - Prob. 16QCh. 10 - Prob. 17QCh. 10 - The conductivity of silicon is enhanced by doping....Ch. 10 - Atoms are assumed to touch in closest packed...Ch. 10 - Define critical temperature and critical pressure....Ch. 10 - Prob. 21QCh. 10 - Describe what is meant by a dynamic equilibrium in...Ch. 10 - Prob. 23QCh. 10 - Prob. 24QCh. 10 - Prob. 25QCh. 10 - Prob. 26QCh. 10 - When wet laundry is hung on a clothesline on a...Ch. 10 - You have three covalent compounds with three very...Ch. 10 - Prob. 30QCh. 10 - Compare and contrast the structures of the...Ch. 10 - Silicon carbide (SiC) is an extremely hard...Ch. 10 - A common prank on college campuses is to switch...Ch. 10 - A plot of In (Pvap) versus 1/T (K) is linear with...Ch. 10 - Prob. 36QCh. 10 - Identify the most important types of interparticle...Ch. 10 - Prob. 38ECh. 10 - Predict which substance in each of the following...Ch. 10 - Consider the compounds CI2, HCI. F2, NaF, and HF....Ch. 10 - Prob. 41ECh. 10 - Consider the following electrostatic potential...Ch. 10 - In each of the following groups of substances,...Ch. 10 - Prob. 44ECh. 10 - The shape of the meniscus of water in a glass tube...Ch. 10 - Prob. 46ECh. 10 - Hydrogen peroxide (H2O2) is a syrupy liquid with a...Ch. 10 - Carbon diselenide (CSe2) is a liquid at room...Ch. 10 - X rays from a copper X-ray tube ( = 154 pm) were...Ch. 10 - The second-order diffraction (n = 2) for a gold...Ch. 10 - A topaz crystal has an interplanar spacing (d) of...Ch. 10 - X rays of wavelength 2.63 were used to analyze a...Ch. 10 - Calcium has a cubic closest packed structure as a...Ch. 10 - Nickel has a face-centered cubic unit cell. The...Ch. 10 - A certain form of lead has a cubic closest packed...Ch. 10 - The density of polonium metal is 9.2 g/cm3. If the...Ch. 10 - You are given a small bar of an unknown metal X....Ch. 10 - A metallic solid with atoms in a face-centered...Ch. 10 - Titanium metal has a body-centered cubic unit...Ch. 10 - Barium has a body-centered cubic structure. If the...Ch. 10 - The radius of gold is 144 pm, and the density is...Ch. 10 - The radius of tungsten is 137 pm and the density...Ch. 10 - What fraction of the total volume of a cubic...Ch. 10 - Iron has a density of 7.86 g/cm3 and crystallizes...Ch. 10 - Prob. 65ECh. 10 - Prob. 66ECh. 10 - Selenium is a semiconductor used in photocopying...Ch. 10 - Prob. 68ECh. 10 - Prob. 69ECh. 10 - Prob. 70ECh. 10 - The structures of some common crystalline...Ch. 10 - The unit cell for nickel arsenide is shown below....Ch. 10 - Cobalt fluoride crystallizes in a closest packed...Ch. 10 - The compounds Na2O, CdS, and ZrI4. all can be...Ch. 10 - What is the formula for the compound that...Ch. 10 - Prob. 76ECh. 10 - A certain metal fluoride crystallizes in such a...Ch. 10 - The structure of manganese fluoride can be...Ch. 10 - The unit cell of MgO is shown below l Does MgO...Ch. 10 - In solid KCl the smallest distance between the...Ch. 10 - The CsCl structure is a simple cubic array of...Ch. 10 - MnO has either the NaCI type structure or the CsCI...Ch. 10 - What type of solid will each of the following...Ch. 10 - What type of solid will each of the following...Ch. 10 - The memory metal, nitinol, is an alloy of nickel...Ch. 10 - Superalloys have been made of nickel and aluminum....Ch. 10 - Perovskite is a mineral containing calcium,...Ch. 10 - A mineral crystallizes in a cubic closest packed...Ch. 10 - Materials containing the elements Y, Ba, Cu, and O...Ch. 10 - The structures of another class of ceramic,...Ch. 10 - Plot the following data and determine Hvap for...Ch. 10 - From the following data for liquid nitric acid,...Ch. 10 - In Breckenridge, Colorado, the typical atmospheric...Ch. 10 - The temperature inside a pressure cooker is 115C....Ch. 10 - Diethyl ether (CH3CH2OCH2CH3) was one of the first...Ch. 10 - Mercury is the only metal that is a liquid at room...Ch. 10 - A substance, X, has the following properties:...Ch. 10 - Use the heating-cooling curve below to answer the...Ch. 10 - The molar heat of fusion of sodium metal is 2.60...Ch. 10 - Prob. 100ECh. 10 - What quantity of energy does it take to convert...Ch. 10 - An ice cube tray contains enough water at 22.0C to...Ch. 10 - A 0.250-g chunk of sodium metal is cautiously...Ch. 10 - Prob. 105ECh. 10 - Prob. 106ECh. 10 - Prob. 107ECh. 10 - Prob. 108ECh. 10 - Prob. 109ECh. 10 - Consider the following data for xenon: Triple...Ch. 10 - Prob. 111AECh. 10 - Consider the following formulas for n-pentane and...Ch. 10 - Some of the physical properties of H2O and D2O are...Ch. 10 - Rationalize the following boiling points:Ch. 10 - Consider the following vapor pressure versus...Ch. 10 - Consider the following enthalpy changes:...Ch. 10 - Consider the following data for an unknown...Ch. 10 - Consider the data for substance X given in...Ch. 10 - Prob. 119AECh. 10 - Boron nitride (BN) exists in two forms. The first...Ch. 10 - Prob. 121AECh. 10 - Argon has a cubic closest packed structure as a...Ch. 10 - Prob. 123AECh. 10 - A 20.0-g sample of ice at 10.0C is mixed with...Ch. 10 - Prob. 125AECh. 10 - Carbon tetrachloride. CCl4, has a vapor pressure...Ch. 10 - A special vessel (see Fig. 10.45) contains ice and...Ch. 10 - Prob. 128AECh. 10 - In regions with dry climates, evaporative coolers...Ch. 10 - Which of the following compound(s) exhibit only...Ch. 10 - Which of the following statements about...Ch. 10 - Prob. 133CWPCh. 10 - Aluminum has an atomic radius of 143 pm and forms...Ch. 10 - Pyrolusite is a mineral containing manganese ions...Ch. 10 - The structure of the compound K2O is best...Ch. 10 - Prob. 137CWPCh. 10 - Some ice cubes at 0c with a total mass of 403 g...Ch. 10 - The enthalpy of vaporization for acetone is 32.0...Ch. 10 - Prob. 140CWPCh. 10 - When I mole of benzene is vaporized at a constant...Ch. 10 - You and a friend each synthesize a compound with...Ch. 10 - Prob. 143CPCh. 10 - Prob. 144CPCh. 10 - Consider two different organic compounds, each...Ch. 10 - Rationalize the differences in physical properties...Ch. 10 - Prob. 147CPCh. 10 - Some ionic compounds contain a mixture of...Ch. 10 - Some ionic compounds contain a mixture of...Ch. 10 - Spinel is a mineral that contains 37.9% aluminum,...Ch. 10 - Mn crystallizes in the same type of cubic unit...Ch. 10 - You are asked to help set up a historical display...Ch. 10 - Some water is placed in a sealed glass container...Ch. 10 - The molar enthalpy of vaporization of water at 373...Ch. 10 - Prob. 155CPCh. 10 - Rubidium chloride has the sodium chloride...Ch. 10 - Prob. 157IPCh. 10 - A metal burns in air at 600c under high pressure...Ch. 10 - Prob. 159IPCh. 10 - General Zod has sold Lex Luthor what Zod claims to...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- please help fill in the tablearrow_forwardAnswer F pleasearrow_forward4. Refer to the data below to answer the following questions: The octapeptide saralasin is a specific antagonist of angiotensin II. A derivative of saralasin is used therapeutically as an antihypertensive. Amino acid analysis of saralasin show the presence of the following amino acids: Ala, Arg, His, Pro, Sar, Tyr, Val, Val A.Sar is the abbreviation for sarcosine, N-methyl aminoethanoic acid. Draw the structure of sarcosine. B. N-Terminal analysis by the Edman method shows saralasin contains sarcosine at the N-terminus. Partial hydrolysis of saralasin with dilute hydrochloric acid yields the following fragments: Tyr-Val-His Sar-Arg-Val His-Pro-Ala Val-Tyr-Val Arg-Val-Tyr What is the structure of saralasin?arrow_forward
- What is the structure of the DNA backbone?arrow_forwardPLEASE PLEASE PLEASE use hand drawn structures when possarrow_forward. M 1- MATCH each of the following terms to a structure from the list below. There is only one correct structure for each term and structures may be used more than once. Place the letter of the structure in the blank to the left of the corresponding term. A. Sanger dideoxy method C. Watson-Crick B. GAUCGUAAA D. translation E. HOH2C OH OH G. transcription I. AUGGCUGAG 0 K. OPOH2C 0- OH N- H NH2 F. -OPOH2C 0- OH OH H. Maxam-Gilbert method J. replication N L. HOH2C a. b. C. d. e. f. g. B M. AGATCGCTC a pyrimidine nucleoside RNA base sequence with guanine at the 3' end. DNA base sequence with cytosine at the 3' end. a purine nucleoside DNA sequencing method for the human genome 2'-deoxyadenosine 5'-phosphate process by which mRNA directs protein synthesis OH NH2arrow_forward
- Please use hand drawn structures when neededarrow_forwardB. Classify the following amino acid. Atoms other than carbon and hydrogen are labeled. a. acidic b. basic C. neutral C. Consider the following image. Which level of protein structure is shown here? a. primary b. secondary c. tertiary d. quaternary D. Consider the following image. H RH H HR H R HR HR RH Which level of protein structure is shown in the box? a. primary b. secondary R c. tertiary d. quaternary コー Rarrow_forwardBriefly answer three from the followings: a. What are the four structures of the protein? b. Why is the side chain (R) attached to the alpha carbon in the amino acids is important for the function? c. What are the types of amino acids? And how is it depend on the (R) structure? d. Write a reaction to prepare an amino acid. prodarrow_forward
- Answe Answer A and B pleasearrow_forward3. Refer to the data below to answer the following questions: Isoelectric point Amino Acid Arginine 10.76 Glutamic Acid 3.22 Tryptophan 5.89 A. Define isoelectric point. B. The most basic amino acid is C. The most acidic amino acid is sidizo zoarrow_forward3. A gas mixture contains 50 mol% H2 and 50 mol% He. 1.00-L samples of this gas mixture are mixed with variable volumes of O2 (at 0 °C and 1 atm). A spark is introduced to allow the mixture to undergo complete combustion. The final volume is measured at 0 °C and 1 atm. Which graph best depicts the final volume as a function of the volume of added O2? (A) 2.00 1.75 Final Volume, L 1.50 1.25 1.00 0.75 0.50 0.25 0.00 0.00 0.25 0.50 2.00 (B) 1.75 1.50 Final Volume, L 1.25 1.00 0.75 0.50- 0.25 0.00 0.75 1.00 0.00 0.25 Volume O₂ added, L 2 0.50 0.75 1.00 Volume O₂ added, L 2 2.00 2.00 (C) (D) 1.75 1.75 1.50 1.50 Final Volume, L 1.25 1.00 0.75 0.50 Final Volume, L 1.25 1.00 0.75 0.50 0.25 0.25 0.00 0.00 0.00 0.25 0.50 0.75 1.00 0.00 0.25 Volume O₂ added, L 0.50 0.75 1.00 Volume O₂ added, L 2arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning
Unit Cell Chemistry Simple Cubic, Body Centered Cubic, Face Centered Cubic Crystal Lattice Structu; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=HCWwRh5CXYU;License: Standard YouTube License, CC-BY