
Concept explainers
Which pair of elements has the most similar Lewis structures?
a. N and S
b. F and Ar
c. Cl and Ar
d. O and S
Interpretation: The pair of elements that would have the most similar structure is to be determined.
Concept Introduction:
Lewis structure is used to represent the molecules. It is also known as dot structure. In Lewis model, valence electrons are represented as dots.
Answer to Problem 1SAQ
Correct Answer: The most similar Lewis structure is of
Therefore, the correct answer is option (d).
Explanation of Solution
Reason for correct option:
Oxygen

Thus,
Hence, option (d) is correct.
Reasons for incorrect options:
Option (a) is given as
Nitrogen falls in group

So, it is a wrong answer.
Option (b) is given as
Fluorine falls in
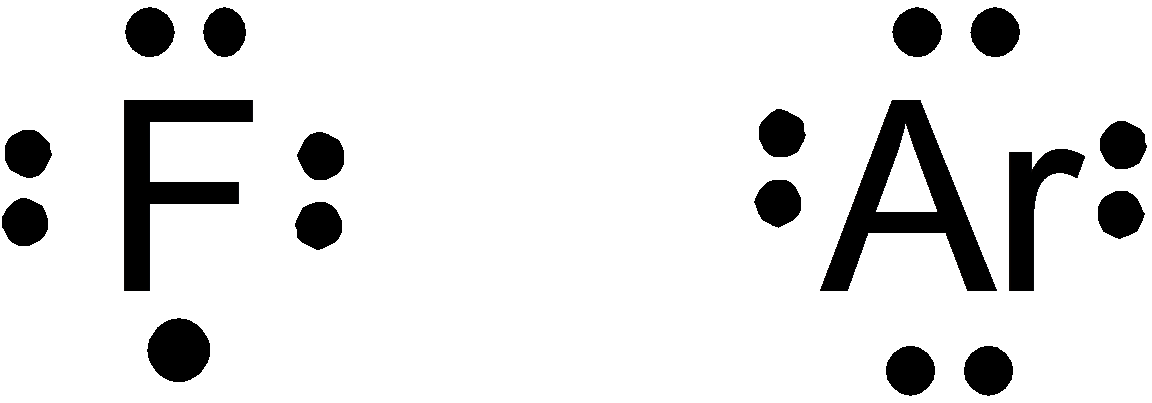
So, it is a wrong answer.
Option (c) is given as
Chlorine falls in group

So, it is a wrong answer.
Hence, options (a), (b) and (c) are incorrect.
Want to see more full solutions like this?
Chapter 10 Solutions
INTRODUCTORY CHEMISTRY-W/SEL.SOLN.MAN.
- Why are normal electrode potentials also called relative electrode potentials?arrow_forwardEasily differentiate between electrochemical potential and Galvani potential.arrow_forwardConstruct a molecular orbital diagram for carbon monoxide. Identify the relevant point group,include all of the appropriate symmetry labels and pictures, and fill in the electrons. Make sure toaccount for the difference in electronegativity between C and O. Hint: CO is substantiallyisoelectronic to N2. (PLEASE DRAW THE ENTIRE MO DIAGRAM!!!)arrow_forward

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





