
(a)
Interpretation:
The symmetry elements and point group of staggered
Concept introduction:
A symmetry operation is defined as an action on an object to reproduce an arrangement that is identical to its original spatial arrangement. The spatial arrangement of the object remains identical after a symmetry operation. The point of reference through which a symmetry operation takes place is termed as a symmetry element.
(a)
Answer to Problem 10A.1P
The symmetry elements of staggered
The staggered
Explanation of Solution
The staggered form of ethane
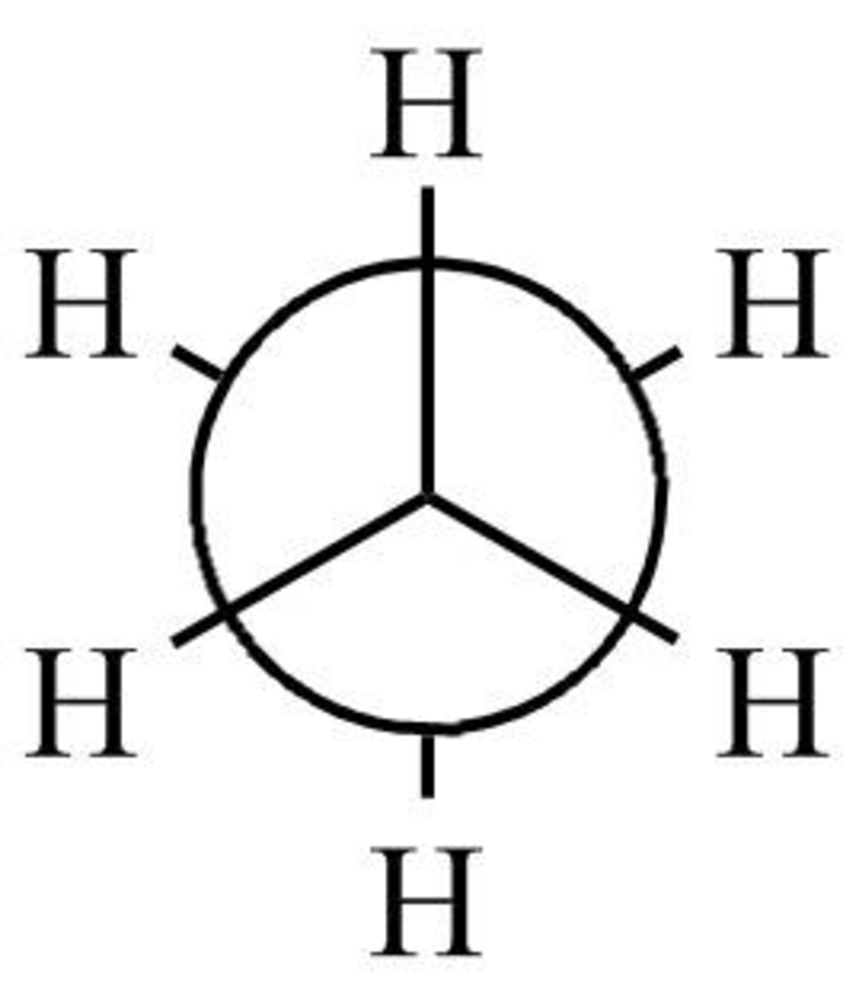
Figure 1
The staggered
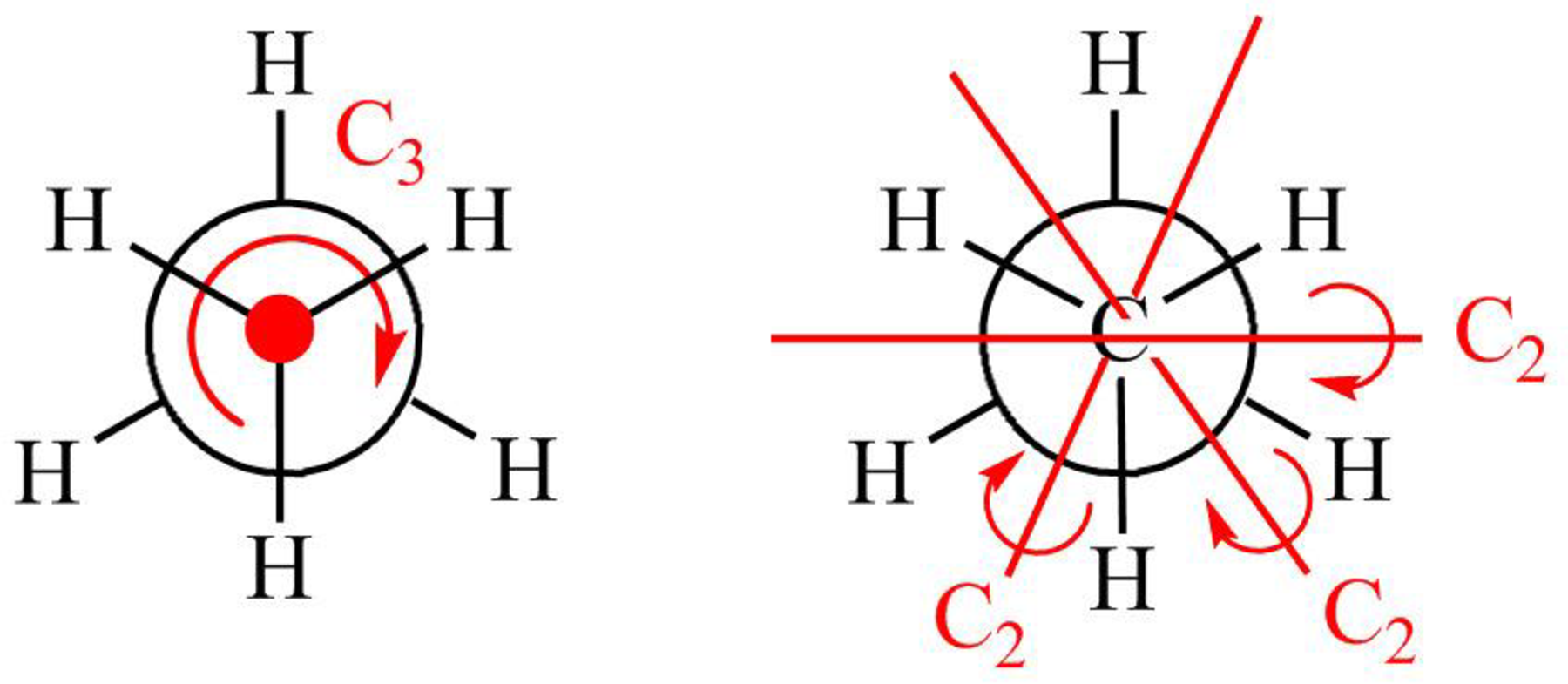
Figure 2
The symmetry elements of staggered
The molecules which have point group
The molecules which do not have any reflective symmetry elements (plane) but may have rotational axis are said to be as a chiral molecule. The staggered
(b)
Interpretation:
The symmetry elements and point group of chair and boat cyclohexane have to be stated. Whether the molecule is polar or not has to be stated. Whether the molecule is chiral or not has to be stated
Concept introduction:
As mentioned in the concept introduction in part (a).
(b)
Answer to Problem 10A.1P
The symmetry elements of the chair form of cyclohexane are
The chair form of cyclohexane molecule is nonpolar and achiral.
The symmetry elements of the boat form of cyclohexane are
The boat form of cyclohexane molecule is polar and achiral.
Explanation of Solution
The structure of the chair form of cyclohexane is shown below in Figure 3.
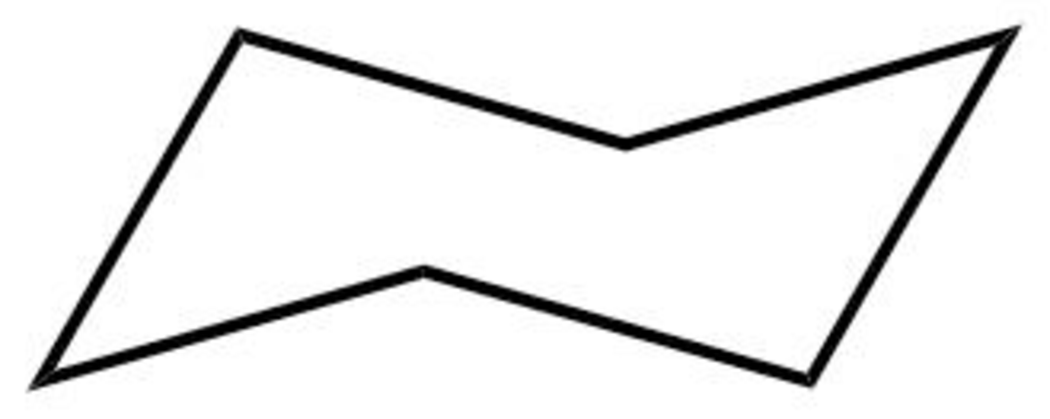
Figure 3
The chair form of cyclohexane has a principal axis
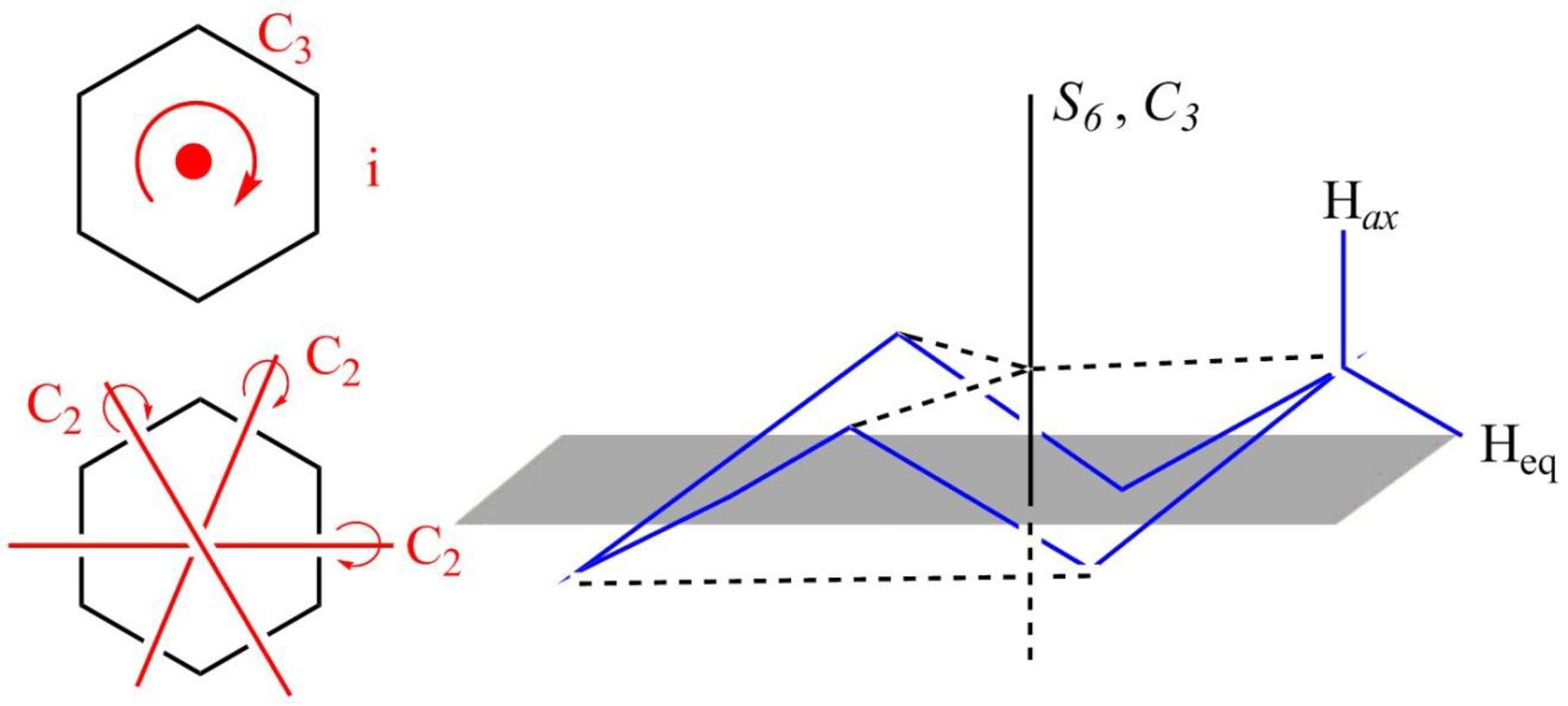
Figure 4
The symmetry elements of the chair form of cyclohexane are
The molecules which have point group
The molecules which do not have any reflective symmetry elements (palne) but may have rotational axis are said to be as a chiral molecule. The chair form of cyclohexane has three
The structure of the boat form of cyclohexane is shown below in Figure 5.
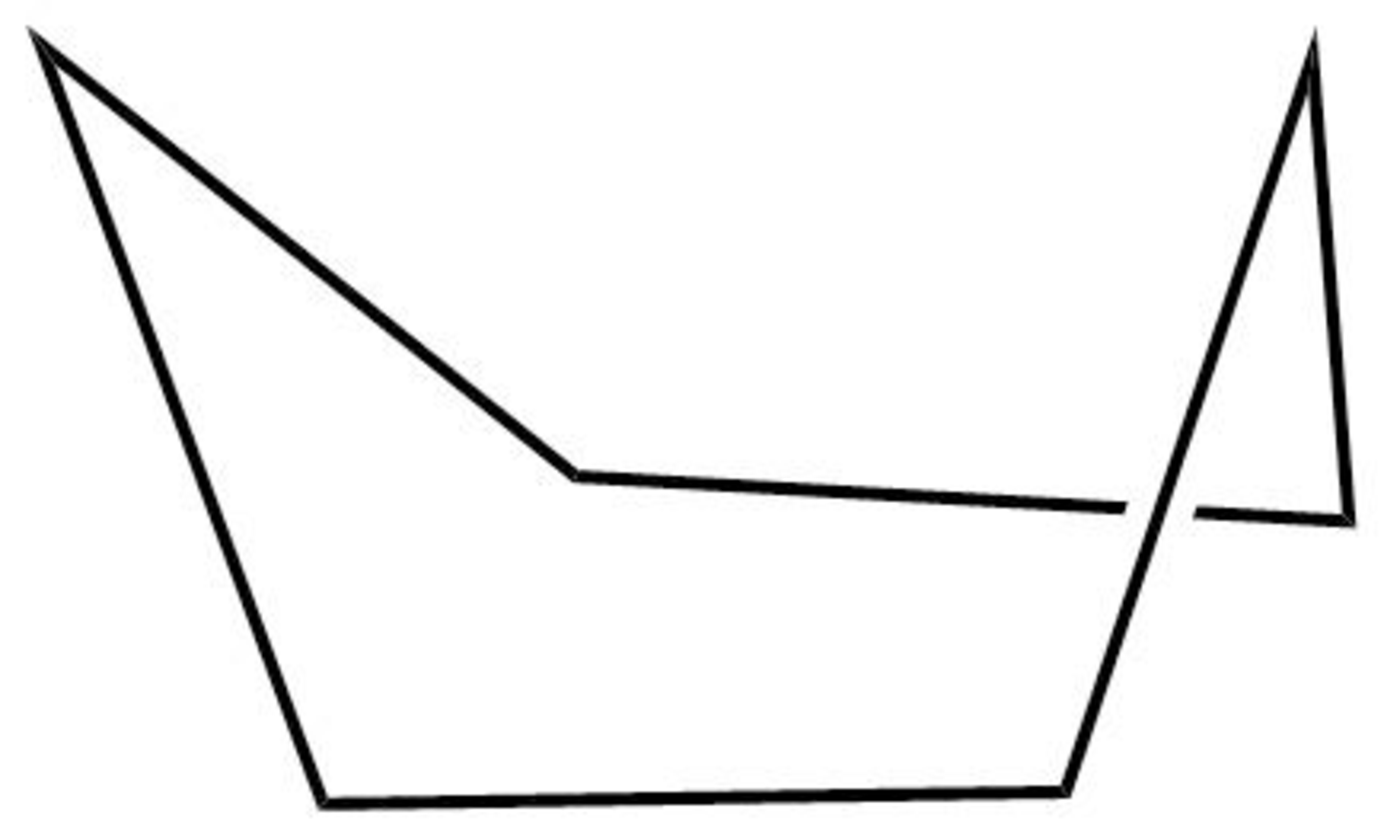
Figure 5
The boat form of cyclohexane has a rotation axis
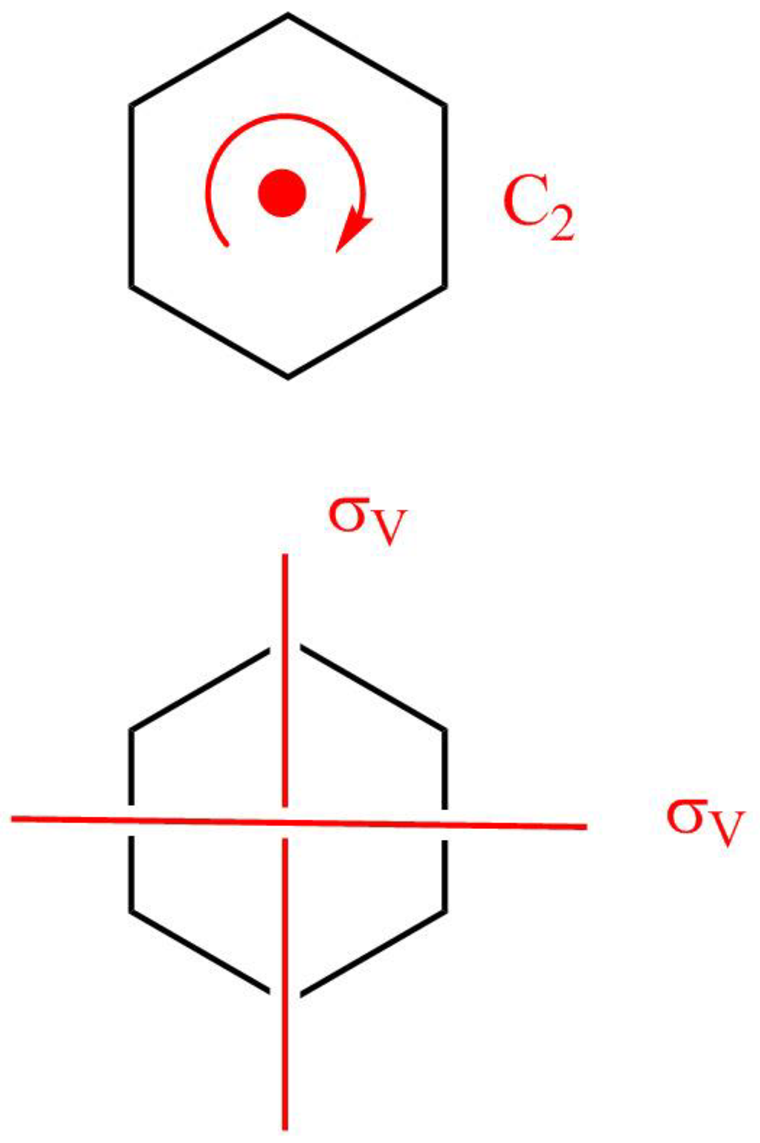
Figure 6
The symmetry elements of the boat form of cyclohexane are
The molecules which have point group
The molecules which do not have any reflective symmetry elements (plane) but may have rotational axis are said to be as a chiral molecule. The boat form of cyclohexane has two
(c)
Interpretation:
The symmetry elements and point group of diborane
Concept introduction:
As mentioned in the concept introduction in part (a).
(c)
Answer to Problem 10A.1P
The symmetry elements of diborane
The diborane
Explanation of Solution
The structure of diborane is shown below in Figure 7.
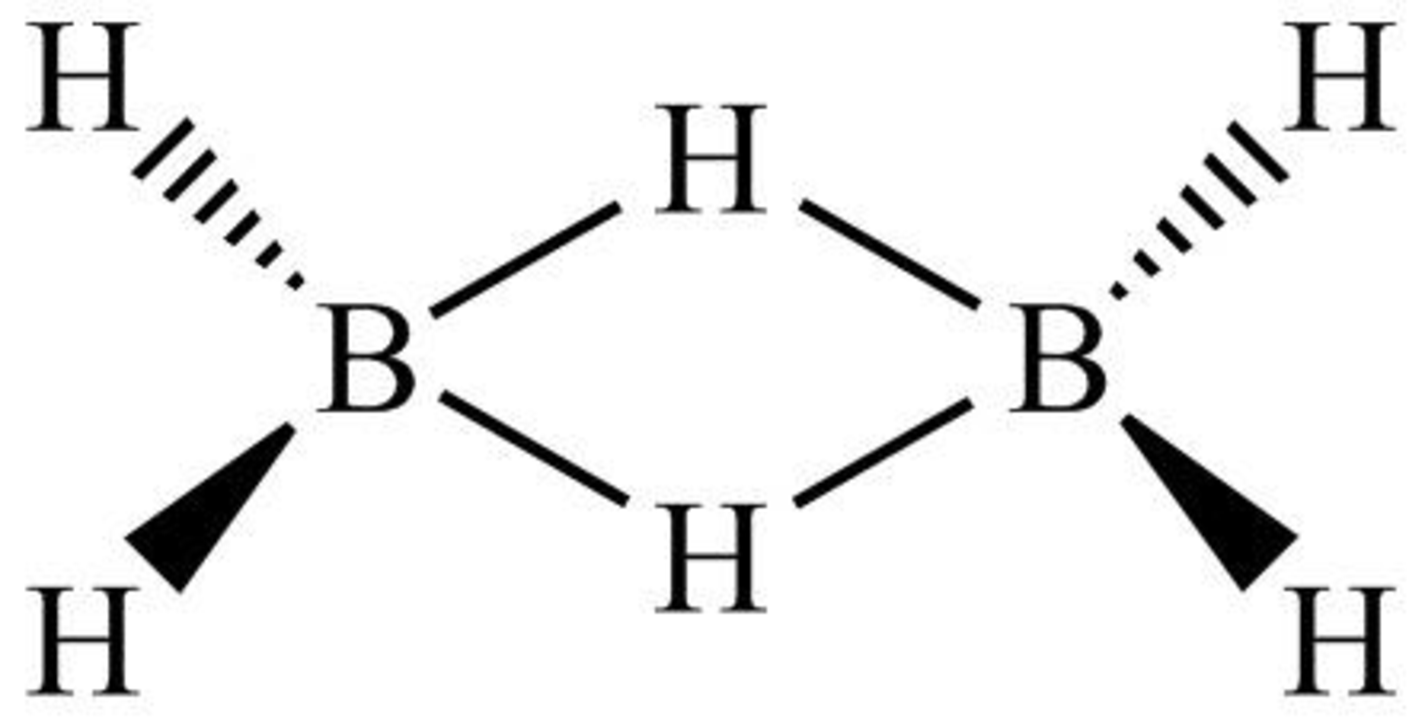
Figure 7
Diborane
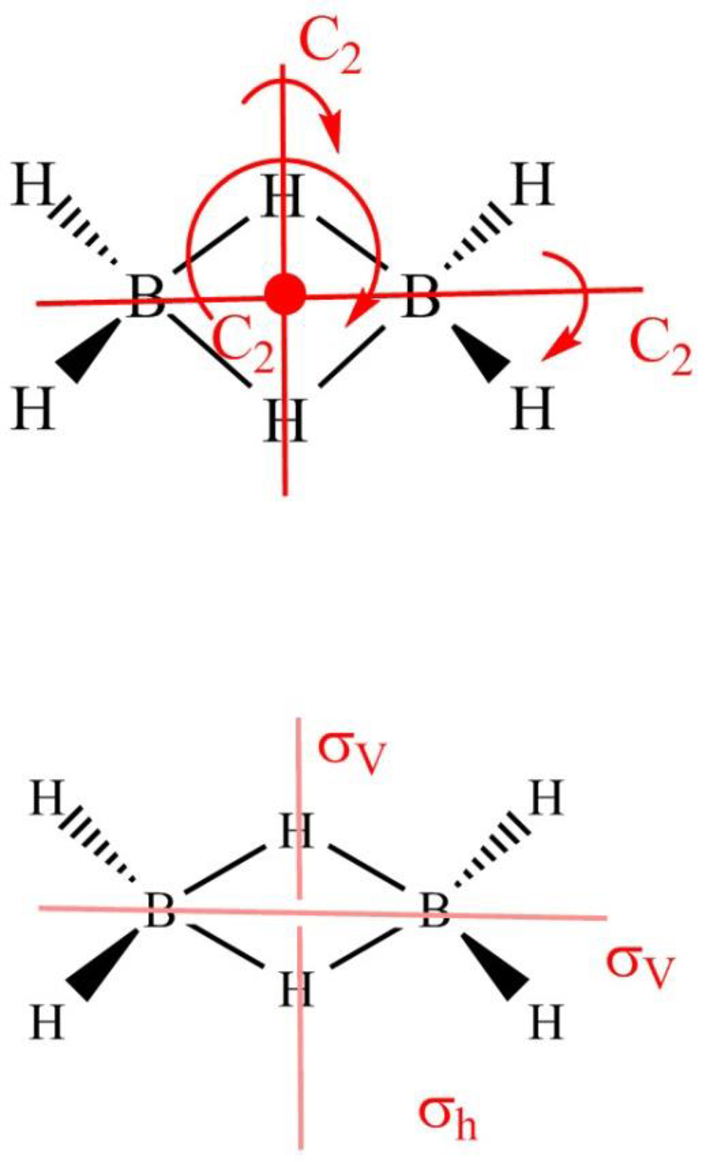
Figure 8
The symmetry elements of diborane
The molecules which have point group
The molecules which do not have any reflective symmetry elements (plane) but may have rotational axis are said to be as a chiral molecule. The diborane
(d)
Interpretation:
The symmetry elements and point group of
Concept introduction:
As mentioned in the concept introduction in part (a).
(d)
Answer to Problem 10A.1P
The symmetry elements of
The
Explanation of Solution
The structure of
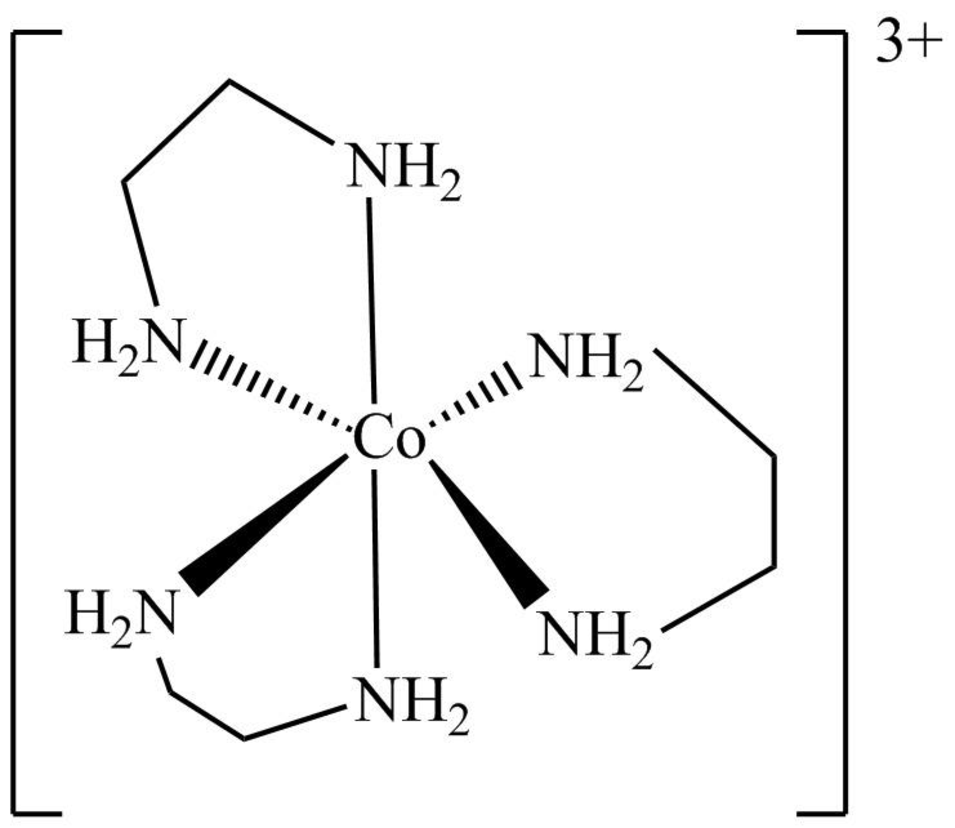
Figure 9
The
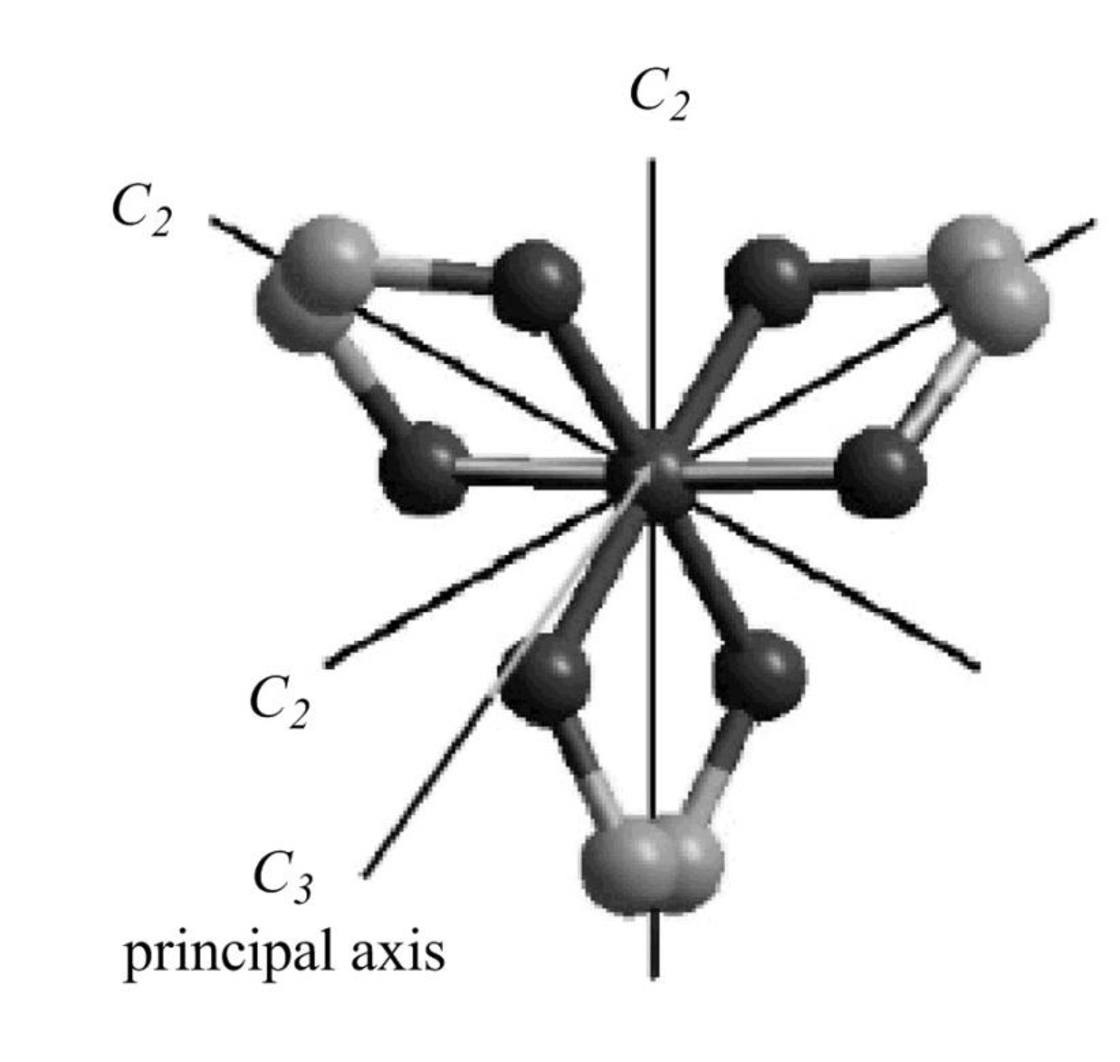
Figure 10
The symmetry elements of
The molecules which have point group
The molecules which do not have any reflective symmetry elements (plane) but may have rotational axis are said to be as a chiral molecule. The
(e)
Interpretation:
The symmetry elements and point group of crown-shaped
Concept introduction:
As mentioned in the concept introduction in part (a).
(e)
Answer to Problem 10A.1P
The symmetry elements of crown-shaped
The crown-shaped
Explanation of Solution
The structure of the crown-shaped
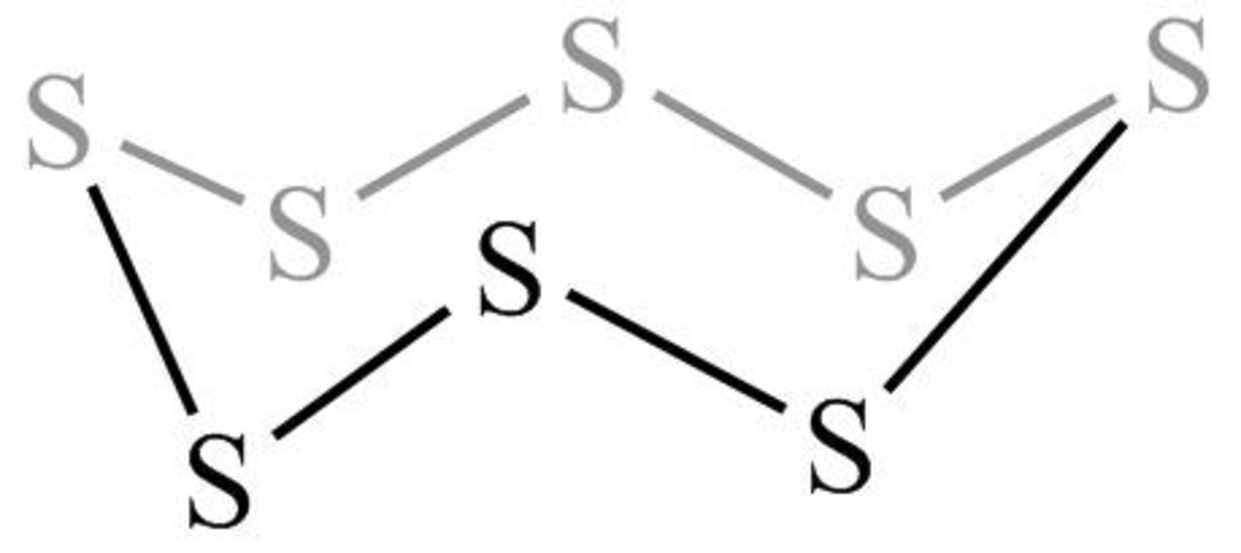
Figure 11
The crown-shaped
The symmetry elements of crown-shaped
The molecules which have point group
The molecules which do not have any reflective symmetry elements (plane) but may have rotational axis are said to be as a chiral molecule. The crown-shaped
Want to see more full solutions like this?
Chapter 10 Solutions
ATKINS' PHYSICAL CHEMISTRY
- Draw the arrow pushing reaction mechanism. DO NOT ANSWER IF YOU WONT DRAW IT. Do not use chat gpt.arrow_forwardComplete the following esterification reaction by drawing the structural formula of the product formed. HOH HO i catalyst catalyst OH HO (product has rum flavor) (product has orange flavor)arrow_forwardThe statements in the tables below are about two different chemical equilibria. The symbols have their usual meaning, for example AG stands for the standard Gibbs free energy of reaction and K stands for the equilibrium constant. In each table, there may be one statement that is faise because it contradicts the other three statements. If you find a false statement, check the box next to t Otherwise, check the "no false statements" box under the table. statement false? AG"1 no false statements: statement false? AG-0 0 InK-0 0 K-1 0 AH-TAS no false statements 2arrow_forward
- Complete the following esterification reactions by drawing the line formulas of the carboxylic acid and alcohol required to form the ester shown. catalyst catalyst catalyst apricot fragrancearrow_forwardShow the saponification products of the following ester: You don't need to draw in the Na+ cation. catalyst, A catalyst, A catalyst, Aarrow_forwardWhat would happen if the carboxylic acid and alcohol groups were on the same molecule? In essence, the molecule reacts with itself. Draw the structure of the products formed in this manner using the reactants below. If two functional groups interact with one another on the same molecule, this is called an “intramolecular" (within one) rather than "intermolecular" (between two or more) attack. OH OH catalyst OH HO catalyst catalyst HO OHarrow_forward
- Q3: Write in the starting alkyl bromide used to form the following products. Include any reactants, reagents, and solvents over the reaction arrow. If more than one step is required, denote separate steps by using 1), 2), 3), etc. H OH racemic OH OH 5 racemicarrow_forwardDraw the Lewis structure of the SO3-O(CH3)2 complex shown in the bottom right of slide 2in lecture 3-3 (“Me” means a CH3 group) – include all valence electron pairs and formal charges.From this structure, should the complex be a stable molecule? Explain.arrow_forwardPredict all organic product(s), including stereoisomers when applicable.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





