
Concept explainers
(a)
Interpretation:
The molecule that has greater dipole moment among
Concept introduction:
The dipole moment arises when there is a separation of charges between two ions or atoms involved in the bond. The dipole moment is a vector quantity and its direction towards the most electronegative atom.
The direction of the dipole moment is represented as follows:
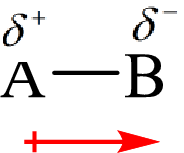
The polar and non-polar molecule can be identified on the basis of the net dipole moment. Polar molecules have non zero value of net dipole moment and the non polar molecules have zero net dipole moment.
(a)
Answer to Problem 10.57P
Explanation of Solution
The shape of
The dipole moment in
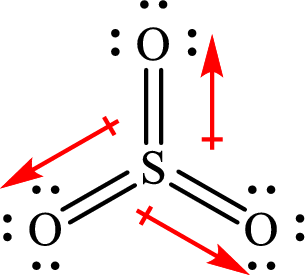
All the
The shape of
The dipole moment in
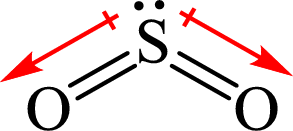
The individual dipole moment does not cancel each other so the value of net dipole moment is not zero and therefore
Hence,
Molecules in which individual dipole moment does not cancel each other result in net non-zero dipole moment and the overall molecule is said to be polar. In
(b)
Interpretation:
The molecule that has greater dipole moment among
Concept introduction:
Electronegativity is the tendency of an atom to attract the shared electrons in the bond towards itself. The more electronegative atom will more attract the bonding electrons towards itself than the less electronegative atom. Therefore the electrons will spend more time with the more electronegative atom than an electropositive atom. The electronegative atom will acquire the partial negative charge and the electropositive atom will acquire a partial positive charge.
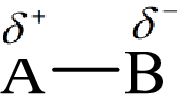
Here, B is the electronegative atom and A is the electropositive atom.
Bond polarity can be estimated by
Here, B is the electronegative atom and A is the electropositive atom.
The greater electronegativity difference between the atoms induces the greater polarity in the bond thus it generates higher dipole moment.
(b)
Answer to Problem 10.57P
Explanation of Solution
The formula to calculate
Substitute
The formula to calculate
Substitute
Bond polarity and dipole moment are directly related to the electronegativity difference. Therefore,
Greater the electronegativity difference between the two bonding atoms more polar will be the bond.
(c)
Interpretation:
The molecule that has greater dipole moment among
Concept introduction:
The dipole moment arises when there is a separation of charges between two ions or atoms involved in the bond. The dipole moment is a vector quantity and its direction towards the most electronegative atom.
The direction of the dipole moment is represented as follows:
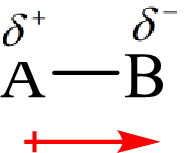
The polar and non-polar molecule can be identified on the basis of the net dipole moment. Polar molecules have non zero value of net dipole moment and the non polar molecules have zero net dipole moment.
(c)
Answer to Problem 10.57P
Explanation of Solution
The shape of
The dipole moment in
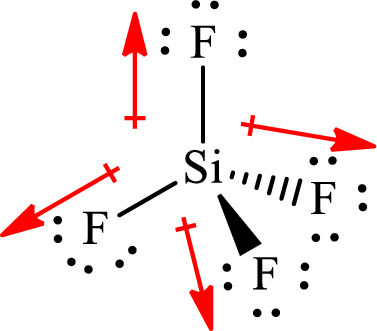
All the
The shape of
The dipole moment in
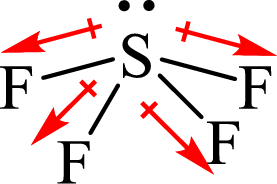
The individual dipole moment does not cancel each other so the value of net dipole moment is not zero and therefore
Hence,
Molecules in which individual dipole moment does not cancel each other result in net non-zero dipole moment and the overall molecule is said to be polar. In
(d)
Interpretation:
The molecule that has greater dipole moment among
Concept introduction:
Electronegativity is the tendency of an atom to attract the shared electrons in the bond towards itself. The more electronegative atom will more attract the bonding electrons towards itself than the less electronegative atom. Therefore the electrons will spend more time with the more electronegative atom than an electropositive atom. The electronegative atom will acquire the partial negative charge and the electropositive atom will acquire a partial positive charge.
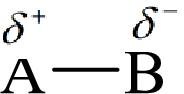
Here, B is the electronegative atom and A is the electropositive atom.
Bond polarity can be estimated by
Here, B is the electronegative atom and A is the electropositive atom.
The greater electronegativity difference between the atoms induces the greater polarity in the bond thus it generates higher dipole moment.
(d)
Answer to Problem 10.57P
Explanation of Solution
The formula to calculate
Substitute
The formula to calculate
Substitute
Both
Greater the electronegativity difference between the two bonding atoms more polar will be the bond.
Want to see more full solutions like this?
Chapter 10 Solutions
Chemistry: The Molecular Nature of Matter and Change - Standalone book
- Which of the following is the most acidic transition metal cation? Group of answer choices Fe3+ Sc3+ Mn4+ Zn2+arrow_forwardBased on the thermodynamics of acetic acid dissociation discussed in Lecture 2-5, what can you conclude about the standard enthalpy change (ΔHo) of acid dissociation for HCl? Group of answer choices You cannot arrive at any of the other three conclusions It is a positive value It is more negative than −0.4 kJ/mol It equals −0.4 kJ/molarrow_forwardPLEASE HELP URGENT!arrow_forward
- Draw the skeletal structure corresponding to the following IUPAC name: 7-isopropyl-3-methyldecanearrow_forwardWhich of the following oxyacids is the weakest? Group of answer choices H2SeO3 Si(OH)4 H2SO4 H3PO4arrow_forwardAdd conditions above and below the arrow that turn the reactant below into the product below in a single transformation. + More... If you need to write reagents above and below the arrow that have complex hydrocarbon groups in them, there is a set of standard abbreviations you can use. More... T H,N NC Datarrow_forward
- Indicate the order of basicity of primary, secondary and tertiary amines.arrow_forward> Classify each of the following molecules as aromatic, antiaromatic, or nonaromatic. Cl Z- N O aromatic O antiaromatic O nonaromatic O aromatic O antiaromatic O nonaromatic O aromatic ○ antiaromatic nonaromaticarrow_forwardPlease help me answer this question. I don't understand how or even if this can happen in a single transformation. Please provide a detailed explanation and a drawing showing how it can happen in a single transformation. Add the necessary reagents and reaction conditions above and below the arrow in this organic reaction. If the products can't be made from the reactant with a single transformation, check the box under the drawing area instead.arrow_forward
- 2) Draw the correct chemical structure (using line-angle drawings / "line structures") from their given IUPAC name: a. (E)-1-chloro-3,4,5-trimethylhex-2-ene b. (Z)-4,5,7-trimethyloct-4-en-2-ol C. (2E,6Z)-4-methylocta-2,6-dienearrow_forwardපිපිම Draw curved arrows to represent the flow of electrons in the reaction on the left Label the reactants on the left as either "Acid" or "Base" (iii) Decide which direction the equilibrium arrows will point in each reaction, based on the given pk, values (a) + H-O H 3-H + (c) H" H + H****H 000 44-00 NH₂ (e) i Дон OH Ө NHarrow_forward3) Label the configuration in each of the following alkenes as E, Z, or N/A (for non-stereogenic centers). 00 E 000 N/A E Br N/A N/A (g) E N/A OH E (b) Oz N/A Br (d) 00 E Z N/A E (f) Oz N/A E (h) Z N/Aarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





