
Concept explainers
The reaction between reactant A (blue spheres) and reactant B (red spheres) is shown in the
following diagram
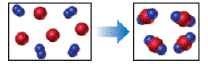
Based on this diagram, which equation best describes the reaction? [Section 3.1D
b.
c.
d.
To determine: The equation that best describes the reaction shown in the given diagram.
Answer to Problem 1E
Solution: The equation (a)
Explanation of Solution
Given
The reaction diagram,
Figure 1 The blue spheres represent the reactant
The red spheres represent the reactant
As depicted in the given diagram, A or blue spheres are present as diatomic molecules
4 molecules of A,
The reaction will be:
Taking coefficients common,
The equation that best describes the depicted chemical reaction is (a)
Want to see more full solutions like this?
Chapter 1 Solutions
Chemistry: The Central Science, Books a la Carte Edition & Modified Mastering Chemistry with Pearson eText -- ValuePack Access Card Package
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
Microbiology: An Introduction
Biology: Life on Earth (11th Edition)
Campbell Biology: Concepts & Connections (9th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
The Cosmic Perspective (8th Edition)
- Hi can you please help me solve this problem? thank youarrow_forwardAn electrode process takes place at a metal-solution interface. Indicate the current condition that must be met for Faradaic rectification to occur.arrow_forwardAt a metal-solution interface, an electron is exchanged, and the symmetry factor beta < 0.5 is found in the Butler-Volmer equation. What does this indicate?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





