his graph shows how the vapor pressure of three liquids varies with temperature: vapor pressure, torr 900- 800- 700- 600- 500- 400- 300- 200 100- 0+ 100 110 120 130 temperature, °C - isobutyl alcohol orthoxylene -acetic acid - 140
his graph shows how the vapor pressure of three liquids varies with temperature: vapor pressure, torr 900- 800- 700- 600- 500- 400- 300- 200 100- 0+ 100 110 120 130 temperature, °C - isobutyl alcohol orthoxylene -acetic acid - 140
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 1RQ: Define and explain the differences between the following terms. a. law and theory b. theory and...
Related questions
Question

Transcribed Image Text:Use the graph to answer the following questions:
Which liquid is the most volatile?
Which is the least volatile?
What is the normal boiling point of each liquid?
Note: your answer must be within 1°C of the exact
answer to b
Suppose a beaker of orthoxylene is put inside a
sealed tank containing orthoxylene gas at 137.
degree C and 409. torr. After ten minutes, will there
be more liquid in the beaker, less liquid, or the same
amount?
most volatile:
least volatile:
isobutyl alcohol:
orthoxylene:
acetic acid:
more
less
the same
choose one
choose one
°℃
°℃
°℃
X
ŵ
î
S

Transcribed Image Text:This graph shows how the vapor pressure of three liquids varies with temperature:
vapor pressure, torr
900
800
700
600
500
400.
300.
200.
100.
0+
100
110
120
130
temperature, °C
Use the graph to answer the following questions:
Which liquid is the most volatile?
Which is the least volatile?
What is the normal boiling point of each liquid?
Note: your answer must be within 1°C of the exact
answer to be y
- isobutyl alcohol
- orthoxylene
acetic acid
140
most volatile:
least volatile:
isobutyl alcohol:
orthoxylene:
acetic acid:
✓ choose one
isobutyl alcohol
orthoxylene
acetic acid
°℃
°C
Expert Solution
Step 1
The vapor pressure-temperature graph of isobutyl alcohol, acetic acid , and ortho-xylene is given as shown in the given problem.In above graph, the isobutyl alcohol is represented by black line, the acetic acid is represented by blue line, and ortho-xylene is represented by green line.
It is known that the temperature at which the vapor pressure of any liquid becomes equal to the atmospheric pressure, is termed as boiling point. The atmospheric pressure id 760 torr.
The temperature corresponds to the 760 torr is the boiling point of corresponding liquid.
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
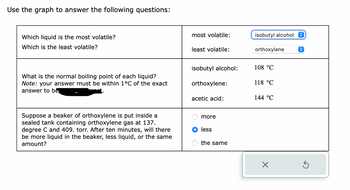
Transcribed Image Text:Use the graph to answer the following questions:
Which liquid is the most volatile?
Which is the least volatile?
What is the normal boiling point of each liquid?
Note: your answer must be within 1°C of the exact
answer to be
Suppose a beaker of orthoxylene is put inside a
sealed tank containing orthoxylene gas at 137.
degree C and 409. torr. After ten minutes, will there
be more liquid in the beaker, less liquid, or the same
amount?
most volatile:
least volatile:
isobutyl alcohol:
orthoxylene:
acetic acid:
more
less
the same
isobutyl alcohol
orthoxylene
108 °C
118 °C
144 °C
×
S
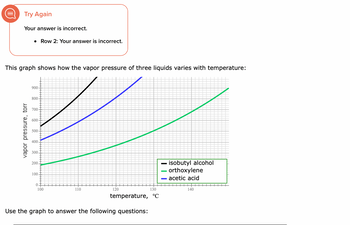
Transcribed Image Text:Try Again
Your answer is incorrect.
• Row 2: Your answer is incorrect.
This graph shows how the vapor pressure of three liquids varies with temperature:
vapor pressure, torr
900
800-
700
600
500-
400
300
200.
100-
0
100
110
120
130
temperature, °C
Use the graph to answer the following questions:
— isobutyl alcohol
- orthoxylene
acetic acid
140
Solution
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9780078021558
Author:
Janice Gorzynski Smith Dr.
Publisher:
McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Elementary Principles of Chemical Processes, Bind…
Chemistry
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY