The enthalpy of evaporation of water is 40.67 kJ>mol. Sunlight striking Earth’s surface supplies 168 W per square meter 11 W = 1 watt = 1 J>s2.) The specific heat capacity of liquid water is 4.184 J>g °C. If the initial surface temperature of a 1.00 square meter patch of ocean is 26 °C, what is its final temperature after being in sunlight for 12 h, assuming no phase changes and assuming that sunlight penetrates uniformly to depth of 10.0 cm?
Energy transfer
The flow of energy from one region to another region is referred to as energy transfer. Since energy is quantitative; it must be transferred to a body or a material to work or to heat the system.
Molar Specific Heat
Heat capacity is the amount of heat energy absorbed or released by a chemical substance per the change in temperature of that substance. The change in heat is also called enthalpy. The SI unit of heat capacity is Joules per Kelvin, which is (J K-1)
Thermal Properties of Matter
Thermal energy is described as one of the form of heat energy which flows from one body of higher temperature to the other with the lower temperature when these two bodies are placed in contact to each other. Heat is described as the form of energy which is transferred between the two systems or in between the systems and their surrounding by the virtue of difference in temperature. Calorimetry is that branch of science which helps in measuring the changes which are taking place in the heat energy of a given body.
The enthalpy of evaporation of water is 40.67 kJ>mol. Sunlight striking Earth’s surface supplies 168 W per square meter 11 W = 1 watt = 1 J>s2.) The specific heat capacity of liquid water is 4.184 J>g °C. If the initial surface temperature of a 1.00 square meter patch of ocean is 26 °C, what is its final temperature after being in sunlight for 12 h, assuming no phase changes and assuming that sunlight penetrates uniformly to depth of 10.0 cm?
The given values with suitable variables are listed below.
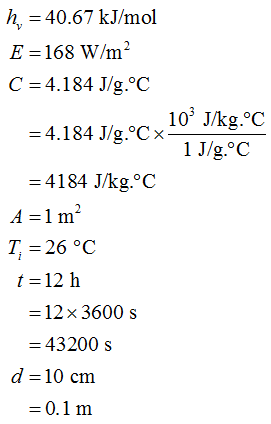
Here, hv denotes the enthalpy of evaporation, E denotes the energy supplied, C denotes the specific heat, A denotes the area, Ti denotes the initial temperature, t denotes the time, and d denotes the depth.
The volume of the water can be determined as,
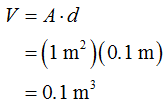
The mass of the water can be determined as,
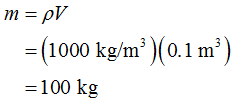
The amount of heat supplied to evaporate water from the ocean can be determined as,
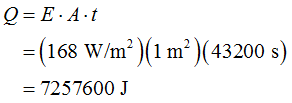
Step by step
Solved in 3 steps with 6 images






