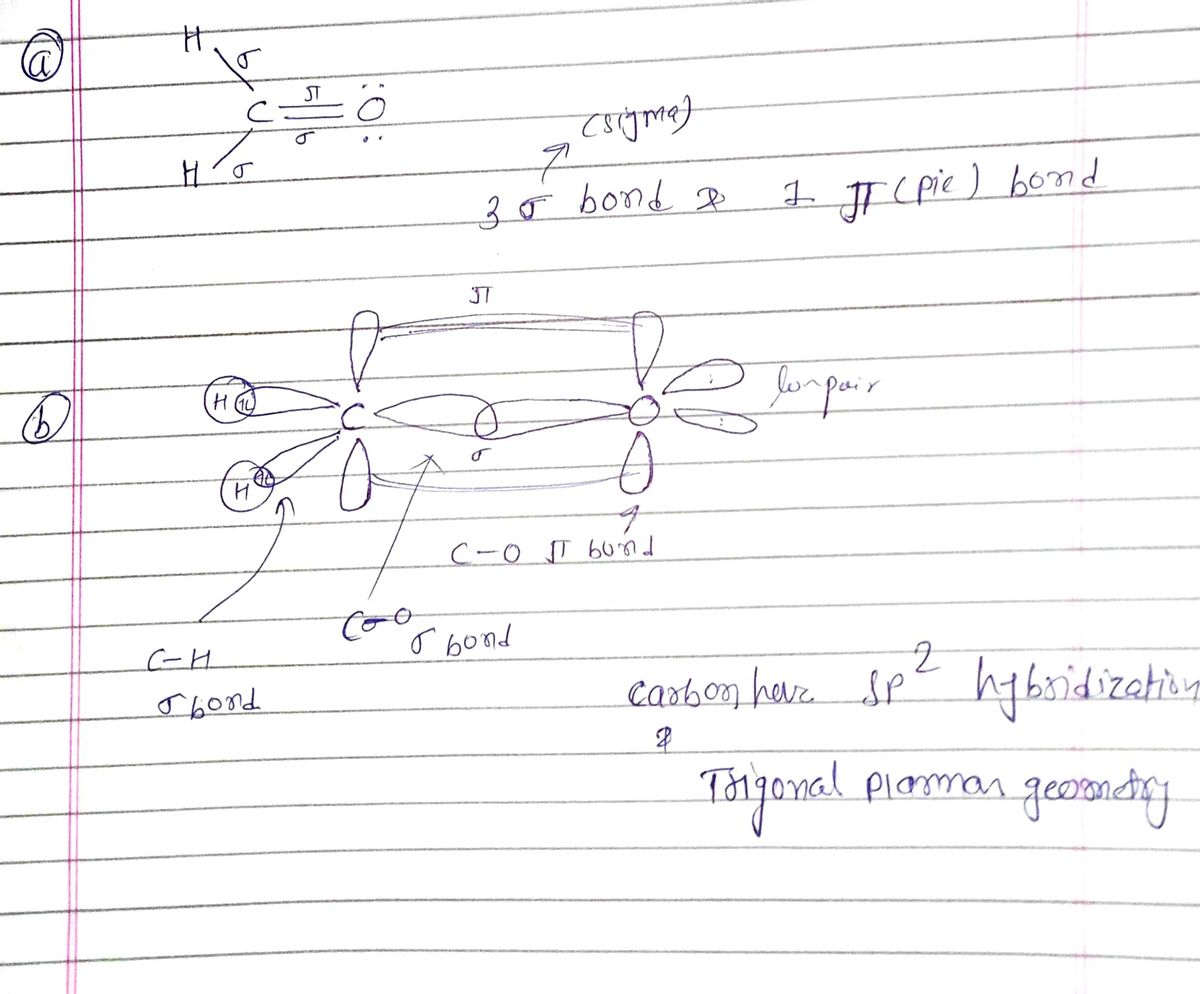
In your Lewis structure for above, label all the bonds as sigma or pi. a) how many sigma and pi bonds are represented? 7. b) for the bonds between C and O in the Lewis structure on the previous page, de the type of orbitals that overlap to form that bond.
In your Lewis structure for above, label all the bonds as sigma or pi. a) how many sigma and pi bonds are represented? 7. b) for the bonds between C and O in the Lewis structure on the previous page, de the type of orbitals that overlap to form that bond.
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 1RQ: Define and explain the differences between the following terms. a. law and theory b. theory and...
Related questions
Question
#7

Transcribed Image Text:## Educational Content on H₂CO Lewis Structures
### For the following: H₂CO
#### State the number of valence electrons and Draw the Lewis Structure:
- **Lewis Structure:**
```
H : C : H
||
O
```
#### State the electron domain geometry and type of hybrid orbital used:
- **Electron Domain Geometry:** Trigonal Planar
#### Orbital Diagrams:
- Orbital Diagram of Central Atom Alone, (C):
- Orbital Diagram of Central Atom in Molecule:
*Note: The aforementioned sections seem to be placeholders for diagrams that are not provided in the image.*
**Questions:**
7. In your Lewis structure for H₂CO above, label all the bonds as sigma (σ) or pi (π).
- **a) How many sigma and pi bonds are represented?**
**Additional Question:**
- For the bonds between C and O in the Lewis structure on the previous page, describe the type of orbitals that overlap to form that bond.
*Note: The image shows content likely intended for students learning about molecular geometry, hybridization, and bonding in formaldehyde (H₂CO).*
Expert Solution
Step 1
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9780078021558
Author:
Janice Gorzynski Smith Dr.
Publisher:
McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Elementary Principles of Chemical Processes, Bind…
Chemistry
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY