Diethyl ether has a freezing point of -116.2 degrees C and a Kf of 1.79 degrees C/m. What is the freezing point of a 1.25 m solution of SO3 in diethyl ether?
Diethyl ether has a freezing point of -116.2 degrees C and a Kf of 1.79 degrees C/m. What is the freezing point of a 1.25 m solution of SO3 in diethyl ether?
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 1RQ: Define and explain the differences between the following terms. a. law and theory b. theory and...
Related questions
Question
Diethyl ether has a freezing point of -116.2 degrees C and a Kf of 1.79 degrees C/m. What is the freezing point of a 1.25 m solution of SO3 in diethyl ether?
Expert Solution
Step 1
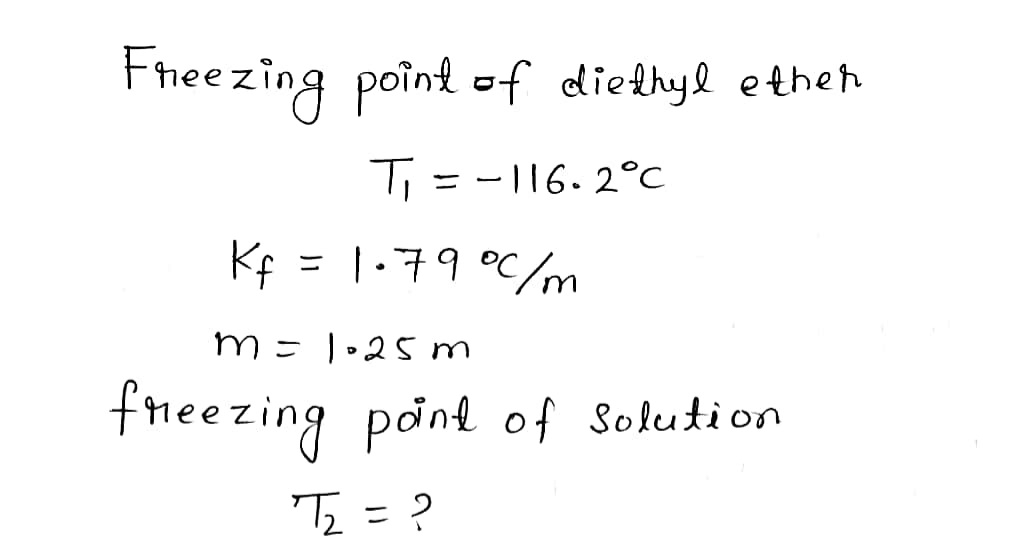
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9780078021558
Author:
Janice Gorzynski Smith Dr.
Publisher:
McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Elementary Principles of Chemical Processes, Bind…
Chemistry
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY
Expert Answers to Latest Homework Questions
Q: Help me answer this please
Q: Suppose the unemployment rate is 10% this month and there are 20 million officially unemployed…
Q: Don't use chat gpt It Chatgpt means downvote
Q: 9. Un cohete es disparado verticalmente hacia arriba desde el suelo. En el instante en que llega a…
Q: E
em
ssion
Use MATLAB's eig command to solve for the eigenvalues of the k - Mw² matrix above.
Then…
Q: Question 3.3 When the air with a pressure of 0.1 MPa and a specific volume of 0.8 m³/kg was provided…
Q: On March 1, 2024, Beldon Corporation purchased land as a factory site for $66,000. An old building…
Q: 5-Methyl-3-nonanone can be prepared in two ways by the conjugate addition of an organocuprate to an…
Q: Please step by step answer and don't use ASI Answer please
Q: Tuke Enterprises provides you with the following information concerning their budget and
actual…
Q: uestion (1 point)
e See section 9.8
A number of reactions can be used to generate common gases on a…
Q: given full explanation and dont AI given solution.....
Q: Use the divergence theorem to evaluate
= z²xi + (v3³.
F(x, y, z) = z²xi +
S
F⚫ds, where
+ tan¯¹(z) )…
Q: Draw the structure(s) of the β-keto ester(s) formed in the Dieckmann reaction of the following…
Q: Molton Inc. made a $60,000 cash expenditure this year (year O). Compute the after-tax
cost if Molton…
Q: Consider an elastic string of length L
==
10 whose ends are held fixed. The string is set in
motion…
Q: January
February
March
Cash
Receipts
$525,000
400,000
450,000
Cash
Payments
$475,000
350,000
525,000…
Q: Which of the following items is consistent with earnings being informative about current performance…
Q: Don't use chat gpt It Chatgpt means downvote please
Q: Beale Management has a noncontributory, defined benefit pension plan. On December 31, 2024 (the end…
Q: A semi-infinite string x > 0 is forced to oscillate by a vertical displacement ŋ = a sin wt
of its…