an be written: СхНу + (x + % у) О2 х СО2 +% у H-О Determine the grams of CO2 output per gram of fuel input for the following alkane fuels: а. Methane — СHA b. Ethane - C2Н6 c. Propane – C3H8
an be written: СхНу + (x + % у) О2 х СО2 +% у H-О Determine the grams of CO2 output per gram of fuel input for the following alkane fuels: а. Methane — СHA b. Ethane - C2Н6 c. Propane – C3H8
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 1RQ: Define and explain the differences between the following terms. a. law and theory b. theory and...
Related questions
Question
![In class, we saw that the chemical reaction for combustion of a hydrocarbon fuel \( C_xH_y \) can be written:
\[ C_xH_y + \left(x + \frac{1}{4} y \right) O_2 \rightarrow x CO_2 + \frac{1}{2} y H_2O \]
Determine the grams of \( CO_2 \) output per gram of fuel input for the following alkane fuels:
a. Methane – \( CH_4 \)
b. Ethane – \( C_2H_6 \)
c. Propane – \( C_3H_8 \)](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Fab5543b4-6299-4fca-ba3a-78b286468977%2F25addc81-fe90-42f6-8b52-0559977892db%2Fl1g4vas_processed.png&w=3840&q=75)
Transcribed Image Text:In class, we saw that the chemical reaction for combustion of a hydrocarbon fuel \( C_xH_y \) can be written:
\[ C_xH_y + \left(x + \frac{1}{4} y \right) O_2 \rightarrow x CO_2 + \frac{1}{2} y H_2O \]
Determine the grams of \( CO_2 \) output per gram of fuel input for the following alkane fuels:
a. Methane – \( CH_4 \)
b. Ethane – \( C_2H_6 \)
c. Propane – \( C_3H_8 \)
Expert Solution
Step 1
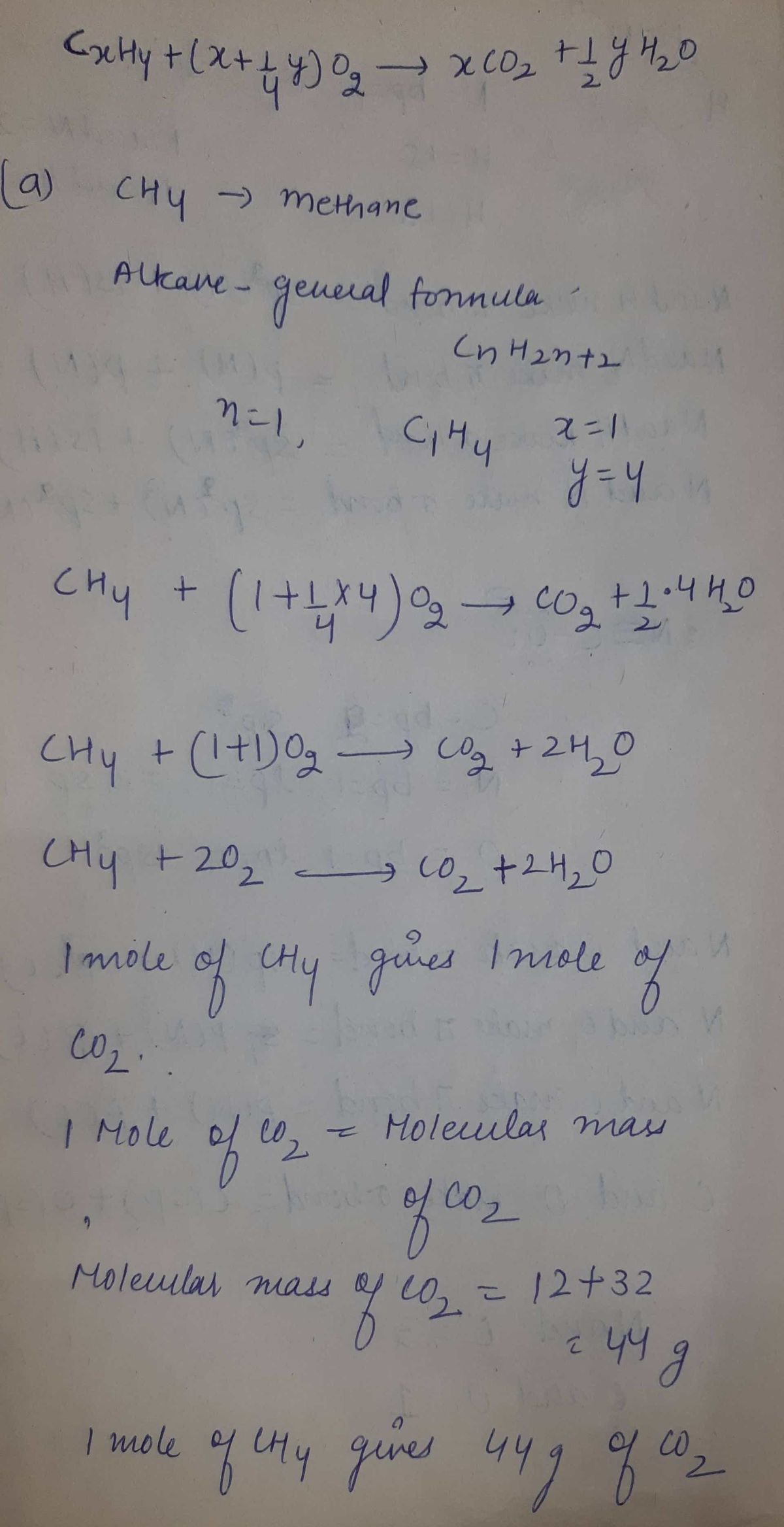
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9780078021558
Author:
Janice Gorzynski Smith Dr.
Publisher:
McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Elementary Principles of Chemical Processes, Bind…
Chemistry
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY