
EBK CHEMISTRY: AN ATOMS FIRST APPROACH
2nd Edition
ISBN: 8220100552236
Author: ZUMDAHL
Publisher: CENGAGE L
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 69E
Given the following data
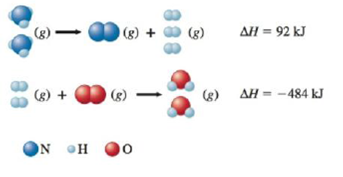
calculate ∆H for the reaction
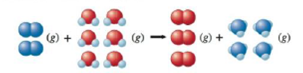
On the basis of the enthalpy change, is this a useful reaction for the synthesis of ammonia?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Consider this molecule:
How many H atoms are in this molecule?
How many different signals could be found in its 1H NMR spectrum?
Note: A multiplet is considered one signal.
For each of the given mass spectrum data, identify whether the compound contains chlorine, bromine, or neither.
Compound
m/z of M* peak
m/z of M
+ 2 peak
ratio of M+ : M
+ 2 peak
Which element is present?
A
122
no M
+ 2 peak
not applicable
(Choose one)
B
78
80
3:1
(Choose one)
C
227
229
1:1
(Choose one)
Show transformation from reactant to product, step by step. *see image
Chapter 7 Solutions
EBK CHEMISTRY: AN ATOMS FIRST APPROACH
Ch. 7 - Define the following terms: potential energy,...Ch. 7 - Consider the following potential energy diagrams...Ch. 7 - What is the first law of thermodynamics? How can a...Ch. 7 - When a gas expands, what is the sign of w? Why?...Ch. 7 - Prob. 5RQCh. 7 - High-quality audio amplifiers generate large...Ch. 7 - Explain how calorimetry works to calculate H or E...Ch. 7 - What is Hesss law? When a reaction is reversed,...Ch. 7 - Define the standard enthalpy of formation. What...Ch. 7 - Prob. 1ALQ
Ch. 7 - Prob. 2ALQCh. 7 - A fire is started in a fireplace by striking a...Ch. 7 - Liquid water turns to ice. Is this process...Ch. 7 - Prob. 5ALQCh. 7 - Prob. 6ALQCh. 7 - Consider 5.5 L of a gas at a pressure of 3.0 atm...Ch. 7 - Explain why oceanfront areas generally have...Ch. 7 - Hesss law is really just another statement of the...Ch. 7 - Prob. 10ALQCh. 7 - Prob. 11QCh. 7 - Prob. 12QCh. 7 - Assuming gasoline is pure C8H18(l), predict the...Ch. 7 - Prob. 14QCh. 7 - The enthalpy change for the reaction...Ch. 7 - For the reaction HgO(s)Hg(l)+12O2(g),H=+90.7KJ: a....Ch. 7 - Prob. 17QCh. 7 - The enthalpy change for a reaction is a state...Ch. 7 - Standard enthalpies of formation are relative...Ch. 7 - The combustion of methane can be represented as...Ch. 7 - Prob. 21QCh. 7 - Prob. 22QCh. 7 - Prob. 23QCh. 7 - Prob. 24QCh. 7 - Prob. 25ECh. 7 - Prob. 26ECh. 7 - Consider the following diagram when answering the...Ch. 7 - Consider the accompanying diagram. Ball A is...Ch. 7 - A gas absorbs 45 kJ of heat and does 29 kJ of...Ch. 7 - A system releases 125 kJ of heat while 104 kJ of...Ch. 7 - Calculate E for each of the following. a. q = 47...Ch. 7 - A system undergoes a process consisting of the...Ch. 7 - If the internal energy of a thermodynamic system...Ch. 7 - Calculate the internal energy change for each of...Ch. 7 - A sample of an ideal gas at 15.0 atm and 10.0 L is...Ch. 7 - Prob. 36ECh. 7 - Consider a mixture of air and gasoline vapor in a...Ch. 7 - As a system increases in volume, it absorbs 52.5 J...Ch. 7 - A balloon filled with 39.1 moles of helium has a...Ch. 7 - Prob. 40ECh. 7 - One of the components of polluted air is NO. It is...Ch. 7 - Prob. 42ECh. 7 - Are the following processes exothermic or...Ch. 7 - Are the following processes exothermic or...Ch. 7 - The overall reaction in a commercial heat pack can...Ch. 7 - Consider the following reaction:...Ch. 7 - Consider the combustion of propane:...Ch. 7 - Consider the following reaction:...Ch. 7 - Prob. 49ECh. 7 - The specific heat capacity of silver is 0.24 J/Cg....Ch. 7 - A 500-g sample of one of the substances listed in...Ch. 7 - Prob. 52ECh. 7 - A 30.0-g sample of water at 280. K is mixed with...Ch. 7 - A biology experiment requires the preparation of a...Ch. 7 - A 5.00-g sample of aluminum pellets (specific heat...Ch. 7 - Hydrogen gives off 120. J/g of energy when burned...Ch. 7 - Prob. 57ECh. 7 - A 110.-g sample of copper (specific heat capacity...Ch. 7 - In a coffee-cup calorimeter, 50.0 mL of 0.100 M...Ch. 7 - In a coffee-cup calorimeter, 100.0 mL of 1.0 M...Ch. 7 - A coffee-cup calorimeter initially contains 125 g...Ch. 7 - In a coffee-cup calorimeter, 1.60 g NH4NO3 is...Ch. 7 - Consider the dissolution of CaCl2:...Ch. 7 - Consider the reaction...Ch. 7 - The heat capacity of a bomb calorimeter was...Ch. 7 - The combustion of 0.1584 g benzoic acid increases...Ch. 7 - The enthalpy of combustion of solid carbon to form...Ch. 7 - Combustion reactions involve reacting a substance...Ch. 7 - Given the following data calculate H for the...Ch. 7 - Given the following data...Ch. 7 - Prob. 71ECh. 7 - Calculate H for the reaction...Ch. 7 - Given the following data...Ch. 7 - Given the following data...Ch. 7 - Give the definition of the standard enthalpy of...Ch. 7 - Write reactions for which the enthalpy change will...Ch. 7 - Prob. 77ECh. 7 - Use the values of Hf in Appendix 4 to calculate H...Ch. 7 - The Ostwald process for the commercial production...Ch. 7 - Calculate H for each of the following reactions...Ch. 7 - The reusable booster rockets of the space shuttle...Ch. 7 - The space shuttle Orbiter utilizes the oxidation...Ch. 7 - Consider the reaction...Ch. 7 - The standard enthalpy of combustion of ethene gas,...Ch. 7 - Water gas is produced from the reaction of steam...Ch. 7 - Prob. 86ECh. 7 - Prob. 87ECh. 7 - Prob. 88ECh. 7 - Some automobiles and buses have been equipped to...Ch. 7 - The complete combustion of acetylene, C2H2(g),...Ch. 7 - Prob. 91AECh. 7 - One way to lose weight is to exercise! Walking...Ch. 7 - Three gas-phase reactions were run in a...Ch. 7 - Nitrogen gas reacts with hydrogen gas to form...Ch. 7 - Combustion of table sugar produces CO2(g) and H2O(...Ch. 7 - Prob. 96AECh. 7 - Consider the following cyclic process carried out...Ch. 7 - Calculate H for the reaction...Ch. 7 - The enthalpy of neutralization for the reaction of...Ch. 7 - Prob. 100AECh. 7 - If a student performs an endothermic reaction in a...Ch. 7 - In a bomb calorimeter, the reaction vessel is...Ch. 7 - The bomb calorimeter in Exercise 102 is filled...Ch. 7 - Prob. 104AECh. 7 - Consider the following equations:...Ch. 7 - Prob. 106AECh. 7 - At 298 K, the standard enthalpies of formation for...Ch. 7 - Prob. 108AECh. 7 - A sample of nickel is heated to 99.8C and placed...Ch. 7 - Quinone is an important type of molecule that is...Ch. 7 - Calculate H for each of the following reactions,...Ch. 7 - Compare your answers from parts a and b of...Ch. 7 - Compare your answer from Exercise 72 of Chapter 3...Ch. 7 - Consider a balloon filled with helium at the...Ch. 7 - Prob. 115CWPCh. 7 - Prob. 116CWPCh. 7 - Prob. 117CWPCh. 7 - A swimming pool, 10.0 m by 4.0 m, is filled with...Ch. 7 - Prob. 119CWPCh. 7 - Calculate H for the reaction...Ch. 7 - Which of the following substances have an enthalpy...Ch. 7 - Consider 2.00 moles of an ideal gas that are taken...Ch. 7 - For the process H2O(l)H2O(g) at 298 K and 1.0 atm,...Ch. 7 - The sun supplies energy at a rate of about 1.0...Ch. 7 - Prob. 125CPCh. 7 - The standard enthalpies of formation for S(g),...Ch. 7 - Use the following standard enthalpies of formation...Ch. 7 - The standard enthalpy of formation for N2H4(g) is...Ch. 7 - The standard enthalpy of formation for NO(g) is...Ch. 7 - A piece of chocolate cake contains about 400...Ch. 7 - You have a l.00-mole sample of water at 30.C and...Ch. 7 - A 500.0-g sample of an element at 195C is dropped...Ch. 7 - A cubic piece of uranium metal (specific heat...Ch. 7 - On Easter Sunday, April 3, 1983, nitric acid...Ch. 7 - Using data from Chapter 2, calculate the change in...Ch. 7 - In Exercise 89 in Chapter 3, the Lewis structures...Ch. 7 - A gaseous hydrocarbon reacts completely with...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2' P17E.6 The oxidation of NO to NO 2 2 NO(g) + O2(g) → 2NO2(g), proceeds by the following mechanism: NO + NO → N₂O₂ k₁ N2O2 NO NO K = N2O2 + O2 → NO2 + NO₂ Ко Verify that application of the steady-state approximation to the intermediate N2O2 results in the rate law d[NO₂] _ 2kk₁[NO][O₂] = dt k+k₁₂[O₂]arrow_forwardPLEASE ANSWER BOTH i) and ii) !!!!arrow_forwardE17E.2(a) The following mechanism has been proposed for the decomposition of ozone in the atmosphere: 03 → 0₂+0 k₁ O₁₂+0 → 03 K →> 2 k₁ Show that if the third step is rate limiting, then the rate law for the decomposition of O3 is second-order in O3 and of order −1 in O̟.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co


Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY