
Concept explainers
Consider the reaction of hydrogen and nitrogen to form ammonia, according to thebalanced equation:
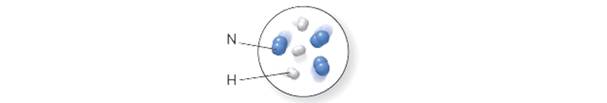
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
General, Organic, & Biological Chemistry
Additional Science Textbook Solutions
Campbell Essential Biology with Physiology (5th Edition)
Biology: Concepts and Investigations
Organic Chemistry (8th Edition)
Biochemistry: Concepts and Connections (2nd Edition)
Chemistry: Structure and Properties (2nd Edition)
- Ammonia can be formed by a direct reaction of nitrogen and hydrogen. N2(g) + 3 H2(g) 2 NH3(g) A tiny portion of the starting mixture is represented by the diagram, where the blue circles represent N and the white circles represent H. Which of these represents the product mixture? For the reaction of the given sample, which of these statements is true? (a) N2 is the limiting reactant. (b) H2 is the limiting reactant. (c) NH, is the limiting reactant. (d) No reactant is limiting: they are present in the correct stoichiometric ratio.arrow_forward4.70 The particulate scale drawing shown depicts the products of a reaction between H2 and O2 molecules. (a) Draw a similar representation for the reactants that must have been present before the reaction took place. (b) Write a balanced chemical equation for the reaction, using the smallest possible whole number coefficients. (c) identify the limiting reactant, and explain how the pictures allow you to do so.arrow_forward4.68 The pictures below show a molecular-scale view of a chemical reaction between the compounds AB2 and B2. (Green balls represent B atoms and orange balls are A atoms). The box on the left represents the reactants at the instant of mixing, and the box on the right shows what is left once the reaction has gone to completion. Was there a limiting reactant in this reaction? If so, what was it? Write a balanced chemical equation for this reaction. As usual, your equation should use the smallest possible whole number coefficients for all substances.arrow_forward
- The sugar sucrose, which is present in many fruits and vegetables, reacts in the presence of certain yeast enzymes to produce ethanol and carbon dioxide gas. Balance the following equation for this reaction of sucrose. C12H22O11(aq) + H2O(l) C2H5OH(aq) + CO2(g)arrow_forward4.71 The particulate scale drawing shown depict the products of a reaction between N2 and O2 molecules. (a) Draw a similar representation for the reactants that must have been present before the reaction took place. (b) Write a balanced chemical equation for the reaction, using the smallest possible whole number coefficients. (c) Identify the limiting reactant, and explain how the pictures allow you to do so.arrow_forwardThe reaction of equal molar amounts of benzene, C6H6, and chlorine, Cl2, carried out under special conditions completely consumes the reactants and yields a gas and a clear liquid. Analysis of the liquid shows that it contains 64.03% carbon, 4.48% hydrogen, and 31.49% chlorine, and has a molar mass of 112.5 g/mol. Write the balanced equation for this reaction.arrow_forward
- 3.75 The following pictures show a molecular-scale view of a chemical reaction between the compounds AB2 and B2. (A atoms are shown in blue and B atoms in white). The box on the left represents the reactants at the instant of mixing, and the box on the right shows what is left once the reac- tion has gone to completion. Write a balanced chemical equation for this reaction. As usual, your equation should use the smallest possible whole number coefficients for all substances.arrow_forwardHigh cost and limited availability of a reactant often dictate which reactant is limiting in a particular process. Identify the limiting reactant when the reactions below are run, and come up with a reason to support your decision. a. Burning charcoal on a grill: C(s)+O2(g)CO2(g) b. Burning a chunk of Mg in water: Mg(s)+2H2O(l)Mg(OH)2(aq)+H2(g) c. The Haber process of ammonia production: 3H2(g)+N2(g)2NH3(g)arrow_forward4.69 The pictures below show a molecular-scale view of a chemical reaction between H2 and CO to produce methanol, CH3OH. The box on the left represents the reactants at the instant of mixing, and the box on the right shows what is left once the reaction has gone to completion. Was there a limiting reactant in this reaction? If so, what was it? Write a balanced chemical equation for this reaction. As usual, your equation should use the smallest possible whole number coefficients for all substances.arrow_forward
- Using solid circles for H atoms and open circles for O atoms, make a drawing that shows the molecular level representation for the balanced equation of H2 and O2 reacting to form H2O.arrow_forwardThe pictures below show a molecular-scale view of a chemical reaction between H2 and CO to produce methanol, CH3OH The box on the left represents the reactants at the instant of mixing, and the box on the right shows what is left once the reaction has gone to completion. D Was there a limiting reactant in this reaction? If so, what was it? Write a balanced chemical equation for this reaction. As usual, your equation should use the smallest possible whole number coefficients for all substances.arrow_forwardAluminum carbide, Al4C3, reacts with water to produce methane, CH4. Al4C3(s) + 12 H2O(l) 4 Al(OH)3(s) + 3 CH4(g) 1. If 125 g of aluminum carbide is decomposed, what is the theoretical yield of methane? (a) 4.64 g (b) 13.9 g (c) 415 g (d) 154 garrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





