
EBK PHYSICS FOR SCIENTISTS & ENGINEERS
5th Edition
ISBN: 9780134296074
Author: GIANCOLI
Publisher: VST
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 26P
(II) Two masses mA = 2.0 kg and mB = 5.0 kg are on inclines and are connected together by a string as shown in Fig. 5–37. The coefficient of kinetic friction between each mass and its incline is μk = 0.30. If mA moves up, and mB moves down, determine their acceleration.
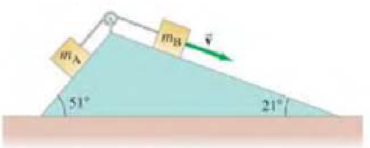
FIGURE 5–37 Problem 28.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
2. A stone is dropped into a pool of water causing ripple to spread out. After 10 s
the circumference of the ripple is 20 m. Calculate the velocity of the wave.
10. Imagine you have a system in which you have 54 grams of ice. You can melt this
ice and then vaporize it all at 0 C. The melting and vaporization are done reversibly
into a balloon held at a pressure of 0.250 bar. Here are some facts about water you
may wish to know. The density of liquid water at 0 C is 1 g/cm³. The density of ice at 0
C is 0.917 g/cm³. The enthalpy of vaporization of liquid water is 2.496 kJ/gram and the
enthalpy of fusion of solid water is 333.55 J/gram.
A. How much energy does the ice absorb as heat when it melts?
B. How much work is involved in melting the ice?
C. What is the total change in energy for melting the ice?
D. What is the enthalpy change for melting the ice?
E. What is the entropy change for melting the ice?
F. What is the change in Helmholtz energy for melting the ice?
G. What is the change in Gibbs energy for melting the ice?
In the figure Q = 5.7 nC and all other quantities are accurate to 2 significant figures. What is the magnitude of the force on the charge Q? (k = 1/4πε 0 = 8.99 × 109 N · m2/C2)
Chapter 5 Solutions
EBK PHYSICS FOR SCIENTISTS & ENGINEERS
Ch. 5.1 - If s = 0.40 and mg = 20 N, what minimum force F...Ch. 5.1 - Prob. 1BECh. 5.2 - Prob. 1CECh. 5.2 - If the radius is doubled to 1.20m but the period...Ch. 5.3 - Prob. 1EECh. 5.3 - A rider on a Ferris wheel moves in a vertical...Ch. 5.4 - Prob. 1GECh. 5.4 - Can a heavy truck and a small car travel safely at...Ch. 5.5 - When the speed of the race car in Example 516 is...Ch. 5 - A heavy crate rests on the bed of a flatbed truck....
Ch. 5 - A block is given a push so that it slides up a...Ch. 5 - Cross-country skiers prefer their skis to have a...Ch. 5 - Prob. 4QCh. 5 - It is not easy to walk on an icy sidewalk without...Ch. 5 - Why is the stopping distance of a truck much...Ch. 5 - A car rounds a curve at a steady 50 km/h. If it...Ch. 5 - Will the acceleration of a car be the same when a...Ch. 5 - Describe all the forces acting on a child riding a...Ch. 5 - A child on a sled comes flying over the crest of a...Ch. 5 - Sometimes it is said that water is removed from...Ch. 5 - Technical reports often specify only the rpm for...Ch. 5 - A girl is whirling a ball on a string around her...Ch. 5 - The game of tetherball is played with a ball tied...Ch. 5 - Astronauts who spend long periods in outer space...Ch. 5 - A bucket of water can be whirled in a vertical...Ch. 5 - A car maintains a constant speed v as it traverses...Ch. 5 - Why do bicycle riders lean in when rounding a...Ch. 5 - Why do airplanes bank when they turn? How would...Ch. 5 - For a drag force of the form F = bv, what are the...Ch. 5 - Suppose two forces act on an object, one force...Ch. 5 - Prob. 2MCQCh. 5 - Prob. 3MCQCh. 5 - Prob. 4MCQCh. 5 - Prob. 5MCQCh. 5 - Prob. 6MCQCh. 5 - Prob. 7MCQCh. 5 - Prob. 8MCQCh. 5 - Prob. 9MCQCh. 5 - Prob. 12MCQCh. 5 - Prob. 13MCQCh. 5 - Prob. 1PCh. 5 - Prob. 2PCh. 5 - (I) Suppose you are standing on a train...Ch. 5 - (I) The coefficient of static friction between...Ch. 5 - Prob. 5PCh. 5 - Prob. 6PCh. 5 - (II) A car can decelerate at 3.80 m/s2 without...Ch. 5 - Prob. 8PCh. 5 - Prob. 9PCh. 5 - (II) A box is given a push so that it slides...Ch. 5 - (II) A skier moves down a 27 slope at constant...Ch. 5 - (II) A wet bar of soap slides freely down a ramp...Ch. 5 - Prob. 13PCh. 5 - Prob. 14PCh. 5 - Prob. 15PCh. 5 - (II) Police investigators, examining the scene of...Ch. 5 - (II) Piles of snow on slippery roofs can become...Ch. 5 - Prob. 18PCh. 5 - (II) Two crates, of mass 65 kg and 125 kg, are in...Ch. 5 - Prob. 20PCh. 5 - (II) A crate is given an initial speed of 3.0 m/s...Ch. 5 - (II) A flatbed truck is carrying a heavy crate....Ch. 5 - Prob. 23PCh. 5 - Prob. 24PCh. 5 - (II) A package of mass m is dropped vertically...Ch. 5 - (II) Two masses mA = 2.0 kg and mB = 5.0 kg are on...Ch. 5 - Prob. 27PCh. 5 - (II) (a) Suppose the coefficient of kinetic...Ch. 5 - Prob. 29PCh. 5 - (II) For two blocks, connected by a cord and...Ch. 5 - Prob. 31PCh. 5 - (III) A 3.0-kg block sits on top of a 5.0-kg block...Ch. 5 - (III) A 4.0-kg block is stacked on top of a...Ch. 5 - (III) A small block of mass m rests on the rough...Ch. 5 - Prob. 35PCh. 5 - Prob. 36PCh. 5 - Prob. 37PCh. 5 - (I) A jet plane traveling 1890 km/h (525 m/s)...Ch. 5 - Prob. 39PCh. 5 - Prob. 40PCh. 5 - Prob. 41PCh. 5 - (II) How fast (in rpm) must a centrifuge rotate if...Ch. 5 - Prob. 43PCh. 5 - (II) Redo Example 511, precisely this time, by not...Ch. 5 - (II) Highway curves are marked with a suggested...Ch. 5 - Prob. 46PCh. 5 - (II) At what minimum speed must a roller coaster...Ch. 5 - Prob. 48PCh. 5 - Prob. 49PCh. 5 - Prob. 50PCh. 5 - Prob. 51PCh. 5 - Prob. 52PCh. 5 - Prob. 53PCh. 5 - Prob. 54PCh. 5 - Prob. 55PCh. 5 - Prob. 56PCh. 5 - Prob. 57PCh. 5 - (II) Two blocks with masses mA and mB, are...Ch. 5 - Prob. 59PCh. 5 - Prob. 60PCh. 5 - (II) A pilot performs an evasive maneuver by...Ch. 5 - (III) The position of a particle moving in the xy...Ch. 5 - (III) If a curve with a radius of 85 m is properly...Ch. 5 - Prob. 65PCh. 5 - Prob. 67PCh. 5 - Prob. 68PCh. 5 - Prob. 69PCh. 5 - (III) An object of mass m is constrained to move...Ch. 5 - (I) Use dimensional analysis (Section 17) in...Ch. 5 - (II) The terminal velocity of a 3 105 kg raindrop...Ch. 5 - (III) Determine a formula for the position and...Ch. 5 - (III) The drag force on large objects such as...Ch. 5 - (II) An object moving vertically has v=v0at t = 0....Ch. 5 - Prob. 77PCh. 5 - Prob. 78PCh. 5 - (III) A motorboat traveling at a speed of 2.4 m/s...Ch. 5 - A coffee cup on the horizontal dashboard of a car...Ch. 5 - Prob. 81GPCh. 5 - Prob. 82GPCh. 5 - Prob. 83GPCh. 5 - A flat puck (mass M) is revolved in a circle on a...Ch. 5 - In a Rotor-ride at a carnival, people rotate in a...Ch. 5 - Prob. 86GPCh. 5 - Prob. 87GPCh. 5 - The 70.0-kg climber in Fig. 550 is supported in...Ch. 5 - A small mass m is set on the surface of a sphere,...Ch. 5 - Prob. 90GPCh. 5 - Prob. 91GPCh. 5 - Prob. 92GPCh. 5 - Prob. 93GPCh. 5 - Prob. 94GPCh. 5 - Prob. 95GPCh. 5 - A car is heading down a slippery road at a speed...Ch. 5 - Prob. 97GPCh. 5 - A banked curve of radius R in a new highway...Ch. 5 - Earth is not quite an inertial frame. We often...Ch. 5 - Prob. 100GPCh. 5 - Prob. 101GPCh. 5 - A car starts rolling down a 1-in-4 hill (1-in-4...Ch. 5 - The sides of a cone make an angle with the...Ch. 5 - Prob. 104GPCh. 5 - A ball of mass m = 1.0 kg at the end of a thin...Ch. 5 - Prob. 106GP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Choose the best answer to each of the following. Explain your reasoning. How does a but Jupiter differ from Jup...
Cosmic Perspective Fundamentals
17. Anthropologists are interested in locating areas in Africa where fossils 4-8 million years old might be fou...
Campbell Biology: Concepts & Connections (9th Edition)
1. Can the magnitude of the displacement vector be more than the distance traveled? Less than the distance trav...
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
A source of electromagnetic radiation produces infrared light. Which of the following could be the wavelength ...
Chemistry: The Central Science (14th Edition)
20. For each pedigree shown,
a. Identify which simple pattern of hereditary trans-mission (autosomal dominant,...
Genetic Analysis: An Integrated Approach (3rd Edition)
How is migration based on circannual rhythms poorly suited for adaptation to global climate change?
Campbell Biology (11th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- Now add a fourth charged particle, particle 3, with positive charge q3, fixed in the yz-plane at (0,d2,d2). What is the net force F→ on particle 0 due solely to this charge? Express your answer (a vector) using k, q0, q3, d2, i^, j^, and k^. Include only the force caused by particle 3.arrow_forwardFor a tornadoes and hurricanes, which of the following is most critical? an alert a watch a warning a predictionarrow_forwardWhen a warm front advances up and over a cold front, what is it called? front inversion stationary front cold front occlusion warm front occlusionarrow_forward
- 1) Consider two positively charged particles, one of charge q0 (particle 0) fixed at the origin, and another of charge q1 (particle 1) fixed on the y-axis at (0,d1,0). What is the net force F→ on particle 0 due to particle 1? Express your answer (a vector) using any or all of k, q0, q1, d1, i^, j^, and k^. 2) Now add a third, negatively charged, particle, whose charge is −q2− (particle 2). Particle 2 fixed on the y-axis at position (0,d2,0). What is the new net force on particle 0, from particle 1 and particle 2? Express your answer (a vector) using any or all of k, q0, q1, q2, d1, d2, i^, j^, and k^. 3) Particle 0 experiences a repulsion from particle 1 and an attraction toward particle 2. For certain values of d1 and d2, the repulsion and attraction should balance each other, resulting in no net force. For what ratio d1/d2 is there no net force on particle 0? Express your answer in terms of any or all of the following variables: k, q0, q1, q2.arrow_forwardA 85 turn, 10.0 cm diameter coil rotates at an angular velocity of 8.00 rad/s in a 1.35 T field, starting with the normal of the plane of the coil perpendicular to the field. Assume that the positive max emf is reached first. (a) What (in V) is the peak emf? 7.17 V (b) At what time (in s) is the peak emf first reached? 0.196 S (c) At what time (in s) is the emf first at its most negative? 0.589 x s (d) What is the period (in s) of the AC voltage output? 0.785 Sarrow_forwardA bobsled starts at the top of a track as human runners sprint from rest and then jump into the sled. Assume they reach 40 km/h from rest after covering a distance of 50 m over flat ice. a. How much work do they do on themselves and the sled which they are pushing given the fact that there are two men of combined mass 185 kg and the sled with a mass of 200 kg? (If you haven't seen bobsledding, watch youtube to understand better what's going on.) b. After this start, the team races down the track and descends vertically by 200 m. At the finish line the sled crosses with a speed of 55 m/s. How much energy was lost to drag and friction along the way down after the men were in the sled?arrow_forward
- For what type of force is it not possible to define a potential energy expression?arrow_forward10. Imagine you have a system in which you have 54 grams of ice. You can melt this ice and then vaporize it all at 0 C. The melting and vaporization are done reversibly into a balloon held at a pressure of 0.250 bar. Here are some facts about water you may wish to know. The density of liquid water at 0 C is 1 g/cm³. The density of ice at 0 C is 0.917 g/cm³. The enthalpy of vaporization of liquid water is 2.496 kJ/gram and the enthalpy of fusion of solid water is 333.55 J/gram.arrow_forwardConsider 1 mole of supercooled water at -10°C. Calculate the entropy change of the water when the supercooled water freezes at -10°C and 1 atm. Useful data: Cp (ice) = 38 J mol-1 K-1 Cp (water) 75J mol −1 K -1 Afus H (0°C) 6026 J mol −1 Assume Cp (ice) and Cp (water) to be independent of temperature.arrow_forward
- The molar enthalpy of vaporization of benzene at its normal boiling point (80.09°C) is 30.72 kJ/mol. Assuming that AvapH and AvapS stay constant at their values at 80.09°C, calculate the value of AvapG at 75.0°C, 80.09°C, and 85.0°C. Hint: Remember that the liquid and vapor phases will be in equilibrium at the normal boiling point.arrow_forward3. The entropy of an ideal gas is S = Nkg In V. Entropy is a state function rather than a path function, and in this problem, you will show an example of the entropy change for an ideal gas being the same when you go between the same two states by two different pathways. A. Express ASV = S2 (V2) - S₁(V1), the change in entropy upon changing the volume from V₁to V2, at fixed particle number N and energy, U. B. Express ASN = S₂(N₂) - S₁ (N₁), the change in entropy upon changing the particle number from N₁ to N2, at fixed volume V and energy U. C. Write an expression for the entropy change, AS, for a two-step process (V₁, N₁) → (V2, N₁) → (V2, N₂) in which the volume changes first at fixed particle number, then the particle number changes at fixed volume. Again, assume energy is constant.arrow_forwardPlease don't use Chatgpt will upvote and give handwritten solutionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University
University Physics Volume 1PhysicsISBN:9781938168277Author:William Moebs, Samuel J. Ling, Jeff SannyPublisher:OpenStax - Rice University College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning
An Introduction to Physical SciencePhysicsISBN:9781305079137Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar TorresPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College

University Physics Volume 1
Physics
ISBN:9781938168277
Author:William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:OpenStax - Rice University

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

An Introduction to Physical Science
Physics
ISBN:9781305079137
Author:James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning
Gravitational Force (Physics Animation); Author: EarthPen;https://www.youtube.com/watch?v=pxp1Z91S5uQ;License: Standard YouTube License, CC-BY