
(a)
Interpretation:
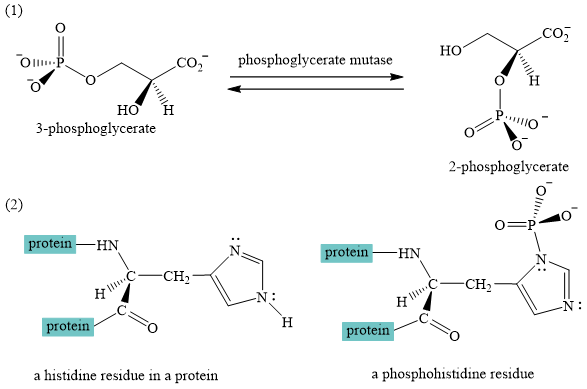
A curved-arrow mechanism for the interconversion of
Concept introduction:
The interconversion of
Answer to Problem 25.35AP
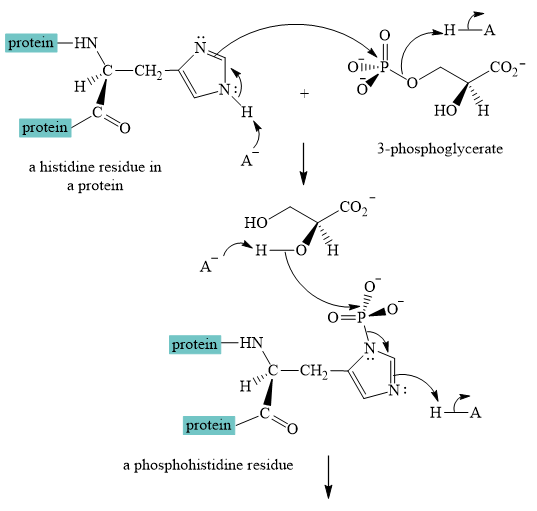
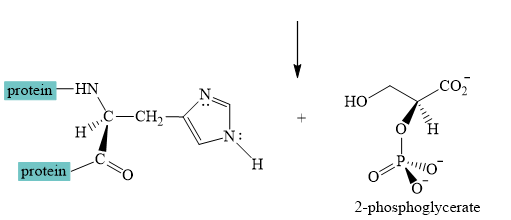
Explanation of Solution
The interconversion equation of
A curved-arrow mechanism for the interconversion of
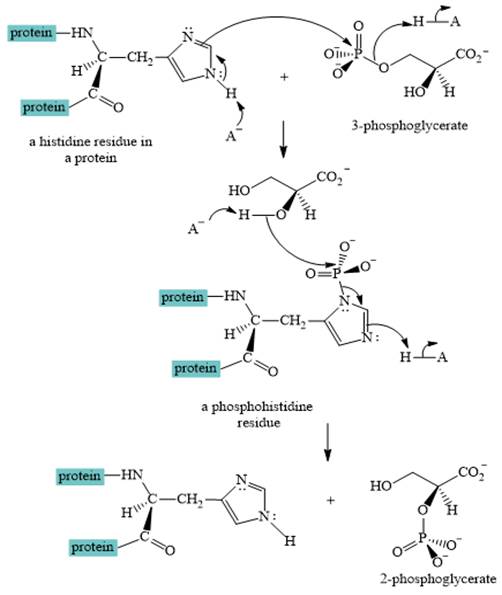
The nitrogen of histidine residue acts as a nucleophile after loss of proton by a base
The two step curved arrow mechanism for the interconversion of
(b)
Interpretation:
If the isotopically chiral
Concept introduction:
The enzyme catalyzed substitution reaction of phosphate ester derivate at phosphate proceeds through
Answer to Problem 25.35AP
If enantiomerically pure
Explanation of Solution
The enzyme catalyzed substitution reaction of phosphate ester derivate at phosphate proceeds through inversion of stereochemistry. The substitution occurs twice with double inversion of the product formed with retention of configuration. Thus, if enantiomerically pure
The interconversion of
Want to see more full solutions like this?
Chapter 25 Solutions
EBK ORGANIC CHEMISTRY STUDY GUIDE AND S
- 2' P17E.6 The oxidation of NO to NO 2 2 NO(g) + O2(g) → 2NO2(g), proceeds by the following mechanism: NO + NO → N₂O₂ k₁ N2O2 NO NO K = N2O2 + O2 → NO2 + NO₂ Ко Verify that application of the steady-state approximation to the intermediate N2O2 results in the rate law d[NO₂] _ 2kk₁[NO][O₂] = dt k+k₁₂[O₂]arrow_forwardPLEASE ANSWER BOTH i) and ii) !!!!arrow_forwardE17E.2(a) The following mechanism has been proposed for the decomposition of ozone in the atmosphere: 03 → 0₂+0 k₁ O₁₂+0 → 03 K →> 2 k₁ Show that if the third step is rate limiting, then the rate law for the decomposition of O3 is second-order in O3 and of order −1 in O̟.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole



